
Market study on telemedicine
October 2018
Third
EU Health
Programme

2
EUROPEAN COMMISSION
Directorate-General for Health and Food Safety
Directorate B — Health systems, medical products and innovation
Unit B.3 — European Reference Networks and Digital Health
E-mail: [email protected]
European Commission
B-1049 Brussels

3
“This document has been prepared for the European Commission however it reflects the
views only of the authors, and the Commission cannot be held responsible for any use
which may be made of the information contained therein.”
More information on the European Union is available on the internet (http://europa.eu).
Luxembourg: Publications Office of the European Union, 2018
ISBN
doi:
© European Union, 2018
Reproduction is authorised provided the source is acknowledged.
Printed in
PRINTED ON ELEMENTAL CHLORINE-FREE BLEACHED PAPER (ECF)
PRINTED ON TOTALLY CHLORINE-FREE BLEACHED PAPER (TCF)
PRINTED ON RECYCLED PAPER
PRINTED ON PROCESS CHLORINE-FREE RECYCLED PAPER (PCF)
Europe Direct is a service to help you find answers
to your questions about the European Union.
Freephone number (*):
00 800 6 7 8 9 10 11
(*) The information given is free, as are most calls (though some operators, phone boxes or hotels
may charge you).

4
LIST OF ABBREVIATIONS
AAL
Ambient Assisted Living
ADI
Integrated Home Care
AI
Artificial Intelligence
AIT
Austrian Institute of Technology
AMA
American Medical Association
ARS
Regional Health Agencies
ATA
American Telemedicine Association
B2B
Business-to-Business
B2C
Business-to-Consumer
BCE
Before the Common Era
BLE
Bluetooth Low Energy
C2B
Consumer-to-Business
CAGR
Compound Annual Growth Rate
CBT
Cognitive Behavioural Therapy
CEA
Cost Effectiveness Analysis
CEF
Connecting Europe Facility
CEMA
Central and Eastern Europe, the Middle East, and Africa
CEN
European Committee for Standardization
CENELEC
European Committee for Electrotechnical Standardization
COPD
Chronic Obstructive Pulmonary Disease
CT
Computed Tomography
CVD
Cardiovascular Disease
DARE
Database of Abstracts of Reviews of Effectiveness
DG CONNECT
Directorate-General for Communications Networks, Content and
Technology
DK
Denmark
DSM
Digital Single Market
EC
European Commission
ECG
Electrocardiogram
ECHI
European Core Health Indicators
ECHIM
European Community Health Indicator Monitoring
EEA
European Economic Area
eHDSI
eHealth Digital Service Infrastructure

5
eHealth
Electronic Health
EHR
Electronic Health Record
ERN
European Reference Network
ETSI
European Telecommunications Standards Institute
EU
European Union
FDA
Food and Drug Administration
FIR
Fonds d’Intervention Régional
GDP
Gross Domestic Product
GDPR
General Data Protection Regulation
GHTF
Global Harmonisation Task Force
GP
General Practitioner
GSMA
GSM Association
HIMSS
Healthcare Information and Management Systems Society
HIT
Health Information Technology
HL7
Health Level Seven International
HR
Human Resources
HTA
Health Technology Assessments
ICER
Incremental Cost-Effectiveness Ratio
ICT
Information and Communication Technology
IHE
Integrating the Health(care) Enterprise
IMDRF
International Medical Device Regulators Forum
IPHS
Integrated Personal Health/care Services
ISO
International Organization for Standardization
IT
Information Technology
ITU-T
ITU Telecommunication Standardization Sector
JA
Joint Action
KPI
Key Performance Indicator
M2M
Machine-to-Machine
MCT
Mobile Cardiac Telemetry
MD
Doctor of Medicine
mHealth
Mobile Health
MHLW
Japanese Ministry of Health, Labour and Welfare
MRI
Magnetic Resonance Imaging
NASA
National Aeronautics and Space Administration
NFC
Near-Field Communication

6
NHS
National Health Service
NST
University Hospital of North Norway
OAD
Home Hospitalisation
P4P
Pay-for-Performance
PCHAlliance
Personal Connected Health Alliance
PDA
Personal Digital Assistant
QALY
Quality Adjusted Life Year
R&D
Research and Development
SDO
Standards Developing Organisation
SIM
Subscriber Identification Module
SIMPHS
Strategic Intelligence Monitor on Personal Health Systems Phase
SIRM
Società Italiana di Radiologia Medica
SME
Small and Medium-sized Enterprises
SWOT
A method to evaluate Strengths, Weaknesses, Opportunities and
Threats
TSB
Technology Strategy Board
UK
United Kingdom
US
United States
WHO
World Health Organization

Market study on telemedicine
Final Report
7
TABLE OF CONTENTS
Printed in ................................................................................................................ 3
List of abbreviations ................................................................................................ 4
Table of contents .................................................................................................... 7
Abstract ................................................................................................................ 9
Executive summary .............................................................................................. 11
Résumé ............................................................................................................... 14
Synthèse ............................................................................................................. 16
Introduction ......................................................................................................... 20
Background ...................................................................................................... 20
Objectives of the study ...................................................................................... 23
Key terms and concepts ........................................................................................ 25
eHealth and mHealth ......................................................................................... 25
Telemedicine ..................................................................................................... 25
Telehealth ........................................................................................................ 25
1. Mapping and categorisation ............................................................................. 27
1.1. Analysis of telemedicine solutions ................................................................. 27
1.1.1. Technical considerations ........................................................................ 28
1.1.2. Geographical distribution........................................................................ 35
1.1.3. Healthcare-context characteristics ........................................................... 40
1.1.4. Analysis of the status of telemedicine solutions ......................................... 46
1.2. Analysis of standards and guidelines ............................................................. 48
1.2.1. Typology of issuing bodies ...................................................................... 48
1.2.2. Geographical spread .............................................................................. 49
1.2.3. Guideline/standard category ................................................................... 49
1.2.4. Medical specialties ................................................................................. 52
2. Market analysis ................................................................................................ 53
2.1. Market fundamentals ................................................................................... 54
2.1.1 Description of the solutions/products that make up the telemedicine market . 54
2.1.2 Geographical distribution of the market .................................................... 55
2.1.3 Focus on three key global markets ........................................................... 58
2.2. Market environment .................................................................................... 61
2.2.1 Market Players ....................................................................................... 61
2.2.2. Supply and demand structure ................................................................. 66
2.2.3. Industry structure and market conditions ................................................. 75
2.3. SWOT analysis of the market ........................................................................ 76

Market study on telemedicine
Final Report
8
2.4. Company profiles ........................................................................................ 80
3. Barriers to access to telemedicine solutions ....................................................... 87
3.1. Identification and analysis of telemedicine framework conditions ...................... 87
3.1.1 Cultural conditions.............................................................................. 89
3.1.2. Regulatory and policy conditions ......................................................... 92
3.1.3. Social security conditions ................................................................... 94
3.1.4. Industrial and technical conditions ........................................................ 96
3.1.5. Knowledge conditions ........................................................................ 97
3.1.6. Financial conditions ............................................................................... 98
3.1.7. Market conditions .................................................................................. 99
3.2. SWOT analysis of the telemedicine framework .............................................. 101
3.3. Policy recommendations ............................................................................ 103
3.3.1. Actions to overcome cultural barriers ..................................................... 103
3.3.2. Actions to overcome regulatory barriers ................................................. 103
3.3.3. Actions to overcome social security barriers ........................................... 104
3.3.4. Actions to overcome industrial/technical barriers ..................................... 104
3.3.5. Actions to overcome knowledge barriers ................................................ 105
3.3.6. Actions to overcome financial barriers .................................................... 105
3.3.7. Actions to overcome market barriers ..................................................... 105
4. Economic analysis ........................................................................................ 107
4.1. Systematic review of cost-effectiveness studies and data collection ................. 108
4.2. Cost-effectiveness of telemedicine solutions ................................................. 111
4.2.1. Baseline scenario – No EU driven efforts to adopt of telemedicine solutions 111
4.2.2. Alternative scenario – EU deployment of telemedicine solutions increase by 5%
rate of adoption ........................................................................................... 114
4.2.3. Description of variables used for economic analysis ................................. 117
4.2.4. Summary of results ............................................................................. 119
4.3. General conclusions................................................................................... 122
Conclusions from the analysis of primary data .................................................. 123
5. Conclusion ................................................................................................... 125
5.1. Main findings ............................................................................................ 125
5.2. Additional considerations ........................................................................... 129

Market study on telemedicine
Final Report
9
ABSTRACT
The aim of the study is to examine the telemedicine market in Europe and to understand
the factors that determine its development. The analysis maps telemedicine applications
and solutions, and applicable technical standards and guidelines; it also describes market
dynamics and potential barriers limiting wider deployment and uptake of telemedicine
solutions. Finally, the study assesses the cost-effectiveness of larger-scale deployment of
telemedicine under current and future market conditions, to provide policy makers with
advice and considerations for wider deployment of telemedicine.
To achieve the study aim, both qualitative and quantitative methods of analysis have
been applied to primary and secondary data. The former includes a survey and interviews
with key stakeholders in the telemedicine market ecosystem. The latter refers to scientific
journals and research reports as well as statistical data.
The study recognises that EU policy makers have undertaken a number of successful
initiatives to facilitate telemedicine adoption. Additional interventions that would support
wider deployment and uptake of telemedicine include: raising public awareness about the
benefits of telemedicine, supporting large-scale projects where telemedicine can be tested
and its benefits assessed, as well as legislative interventions by the EC or MSs to address
some of the barriers for telemedicine adoption in the EU.

Market study on telemedicine
Final Report
10

Market study on telemedicine
Final Report
11
EXECUTIVE SUMMARY
This market study maps European telemedicine solutions and standards, assesses the
current telemedicine market conditions, including barriers, and tests scenarios of the
cost-effectiveness of wider deployment of telemedicine. The data collected, the analysis
performed and the conclusions made can serve to inform policies on telemedicine as a
key area of the Digital Single Market.
First, an analysis of the existing telemedicine solutions and standards/guidelines
in Europe is provided on the basis of a systematic literature review, and in comparison
with the telemedicine market in North America (United States, Canada) and Asia (Japan).
The solutions mapping reveals that telemonitoring and prevention are the
predominant types of intervention for telemedicine solutions, along with
teleconsultation. In terms of longevity, the majority of solutions analysed have been in
use for over five years, which suggests stable demand, potential, and commitment to
invest in this area.
The mapping also shows a concentration of solutions as part of primary care, with
cardiovascular diseases (CVDs), chronic obstructive pulmonary diseases
(COPDs) and diabetes being the most common conditions targeted. It is evident from
the research that a great number of solutions, especially mobile health applications,
target well-being and self-care (non-medical conditions). In terms of solution types,
medical devices and software dominate the market.
Most telemedicine solutions are deployed at the national or regional level, while only
few are in use in multiple MS or outside the EU. This is due to the significant differences
in national regulations and social security schemes, which also incentivised EU policy
makers to take initiatives to promote interoperability between solutions and facilitate
cross-border use. Such initiatives at EU level should remain a priority in the coming
years, to stimulate the development of a vibrant telemedicine market in the EU.
Telemedicine standards and guidelines are found to address mainly technical
requirements. In addition to international bodies, Member States also set their own
national standards, especially to provide precise requirements for telemedicine
solutions related to a given medical specialty. Regarding other types of guideline/rule,
there seems to be good coverage of all relevant domains at present: data protection,
organisational, human resources, ethical and EHR. What may deserve attention in the
future is compatibility between standards, as an enabler for interoperability, when
preparing the deployment of telemedicine services on a large scale.
Second, the study zooms into telemedicine market fundamentals and describes at
length the market environment, culminating with a market SWOT analysis.
It emerges from this part of the market analysis that the uptake of information
technologies in Europe is the main accelerator for telemedicine. The market potential of
telemedicine is demonstrated to be strong and expected to grow at a compound annual
growth rate of 14% in the coming years. The well-being market especially, enabled
by digital technologies, mainly wearables and mobile applications, is also rapidly growing.
Although it appears that demand for telemedicine solution outpaces supply, this
observation should be considered with care, as there are many telemedicine initiatives
and solutions available in the market but hospitals and clinics do not always have the

Market study on telemedicine
Final Report
12
financial resources to adopt the state-of-the-art technology that will allow deployment of
telemedicine services.
Telecommunication companies, ICT tools and electronics manufacturers, device
manufacturers, pharmaceutical industry companies, and start-ups have been identified as
key players in the value chain of the telemedicine market along with patients and health
professionals.
Third, barriers to telemedicine uptake are identified in one of seven categories:
cultural; regulatory and policy; social security; industrial and technical; knowledge;
financial; and market-related. Based on a review of literature, barriers are found to
exist in all European countries but do not affect them to the same degree. Thus, it is
difficult to quantify how the impact of barriers varies across counties. Furthermore, since
telemedicine is a multi-stakeholder market, barriers also affect the players differently
within each country.
Decision-makers should be attentive towards the barriers and pursue actions to overcome
them, in particular: conservatism or resistance to adopting new medical processes,
limited integration between technology and medical practitioner’s procedures, (data
protection) regulations, limited funding/financial incentives and interoperability.
Importantly, uptake of telemedicine solutions across national health systems will also only
be successful if key institutions in the medical community, such as recognised clinics and
hospitals, establish new partnerships. These institutions will only be incentivised to do
so if national decision-makers allow health systems to properly pay the utilisation of the
technology, meaning developing reimbursement schemes for telemedicine utilisation.
Further to this point, it is important to highlight that today, only direct consumer models
have some degree of success, because institutional players cannot pay for or are not
always reimbursed for telemedicine tools and services.
Despite the above barriers, it should be noted that there are a number of areas where EU
and national initiatives have had significant positive impact on telemedicine uptake, for
instance ePrescriptions and the Patient Summary.
Finally, the study offers an economic assessment with the objective of evaluating the
potential benefits of future deployment of telemedicine tools and services across the EU.
This assessment relies on scenario-based analysis using an economic decision model. The
parameters used in the model are based on insight drawn from scientific research
complemented by disease statistics.
In a first step, research databases are examined for evidence of telemedicine cost
effectiveness in medical trials. The main findings of this analysis suggest that
telemedicine is reported to be cost-effective in 73.3% of the cases covered by the
literature, while negative effects account for 5.6% of the selected studies. The remaining
21.3% of the studies analysed present a neutral effect of the use of telemedicine as a
means to save costs. Parameters that have strong impact on the cost-
effectiveness of telemedicine solutions reported by the studies include: distance
between patient and nearest healthcare professional; time required per consultation; cost
of a doctor visit; QALYs; and mortality rate.
In a second step, these cost parameters are used as to assess cost-effectiveness
resulting from wide-scale deployment of telemedicine in Europe, based on
different levels of projected adoption. In this final part, the study examines two scenarios.
Under the first scenario, it is assumed that 18% of health provision, mainly consultation

Market study on telemedicine
Final Report
13
and treatment, take place with the use of telemedicine. The second scenario examines
the impact of an increase in the adoption level by an extra 5% to 23%. In both scenarios,
the costs and benefits of telemedicine are compared to the traditional face-to-face patient
journey to estimate the effect of a wider deployment of telemedicine. It becomes
apparent that the higher the share of telemedicine – the more cost-effective
wide-scale deployment becomes. An increasing share of telemedicine decreases the
total cost of the patient journey, the total consultation time, the total distance travelled
and the rates of mortality, while it increases QALYs gained.
However, this is only a first EE-wide assessment. Policy-makers need to invest in
obtaining more scientific evidence for the efficiency of telemedicine by financing and
monitoring large-scale experiments to assess the impact of a wider deployment. Raising-
awareness (patients, doctors), stimulating integration between stakeholders and
facilitating reimbursement are additional considerations for speeding up adoption and the
realisation of benefits resulting from telemedicine use.

Market study on telemedicine
Final Report
14
RÉSUMÉ
L’objectif de cette étude est d’examiner le marché de la télémédecine en Europe et de
comprendre les facteurs qui déterminent son développement. Cette analyse cartographie
les applications et solutions utilisés par la télémédecine, ainsi que les directives et les
standards techniques en vigueur ; elle décrit également les dynamiques de ce marché et
les obstacles potentiels qui pourraient limiter l’adoption de solutions de télémédecine.
Enfin, cette étude mesure le rapport coût-efficacité d’un déploiement à grande échelle de
la télémédecine dans les conditions de marché actuelles et futures et ainsi mettre à
disposition des décideurs politiques les éléments à considérer pour un plus grand
déploiement de la télémédecine.
Afin d’atteindre les objectifs de cette étude, des méthodes d’analyse qualitative et
quantitative ont été appliquées à des données primaires et secondaires. Les données
primaires sont constituées d’une enquête, ainsi que des entretiens avec les parties
prenantes clés de l’écosystème du marché de la télémédecine. Les données secondaires
font référence à des publications scientifiques et à des rapports de recherche, mais aussi
à des données statistiques.
Les résultats de l’étude indiquent que les décideurs politiques de l’Union Européenne (UE)
ont entrepris un nombre d’initiatives réussies, afin de faciliter l’adoption de la
télémédecine. D’autres interventions pourraient encourager une adoption et un
déploiement plus grands de la télémédecine comme : la sensibilisation du public aux
avantages de la télémédecine, le soutien à des projets d’envergure dans lesquels le
déploiement de la télémédecine peut être testé et ses bénéfices évalués, mais aussi des
interventions législatives par la Commission Européenne ou par les États Membres pour
éliminer certains des obstacles à l’adoption de la télémédecine dans l’UE.

Market study on telemedicine
Final Report
15

Market study on telemedicine
Final Report
16
SYNTHÈSE
Cette étude de marché cartographie les solutions de télémédecine et les standards
européens, évalue les conditions actuelles du marché de la télémédecine, en identifiant
les obstacles et barrières à son déploiement, et modélise des scénarios de type coût-
efficacité d’un déploiement de plus grande ampleur de la télémédecine. Les données
collectées, l’analyse réalisée et les conclusions établies peuvent servir les décideurs
publics pour établir leur politique d’intervention dans le cadre du Marché Unique
Numérique.
Dans un premier temps, une analyse des solutions de télémédecine et des
standards/principes en Europe a été réalisée sur la base d’une revue littéraire
systématique, en comparaison du marché nord-américain (États-Unis, Canada) et
asiatique (Japon).
Le recensement des différentes solutions révèle que le télémonitoring et la
prévention sont les types d’intervention prédominants pour les solutions de
télémédecine, de même que la téléconsultation. En termes de pérennité, la majorité
des solutions analysées sont utilisées depuis plus de cinq ans, ce qui démontre la
stabilité de la demande, le potentiel et la détermination à investir dans ce domaine.
Le recensement montre également une concentration des solutions en matière de soins
primaires et plus particulièrement des maladies cardio-vasculaires, des affections
pulmonaires obstructives primaires et du diabète qui sont des couramment ciblés.
L’analyse met en lumière qu’un grand nombre de solutions, les applications de santé
mobiles particulièrement, ciblent le bien-être et l’auto-traitement (conditions non-
médicales). En termes de types de solutions, les équipements médicaux et les
logiciels dominent le marché.
La plupart des solutions de télémédecine sont déployées au niveau national ou
régional, tandis que très peu sont utilisées dans les États Membres ou en dehors de l’UE.
Ceci est dû aux différences significatives entre les régulations nationales et aux modalités
de prise en charges de la sécurité sociale, ce qui a notamment encouragé les décideurs
politiques de l’UE à prendre des initiatives au niveau de l’UE pour promouvoir
l’interopérabilité entre les solutions et pour faciliter leur utilisation transfrontalière.
Ces initiatives au niveau de l’UE devraient rester une priorité dans les années à venir,
pour stimuler le développement d’un marché de la télémédecine dynamique.
Les standards et directives en matière de télémédecine sont là pour répondre aux
exigences techniques principalement. Au-delà des instances internationales, les États
Membres définissent également leurs propres standards nationaux, particulièrement
lorsqu’il s’agit de fournir des exigences précises pour des solutions de télémédecine
spécifiques à une spécialité médicale. En ce qui concerne les autres types de
directives/règles, il semble qu’il y ait aujourd’hui une bonne couverture de tous les
domaines clés : protection des données, process et ressources humaines, éthique et
dossier électronique du patient. Dans le futur, une attention particulière doit être portée
surla compatibilité entre les standards, en tant que facilitateur de l’interopérabilité,
quand il s’agira de préparer le déploiement des services de télémédecine à grande
échelle.
Dans un deuxième temps, l’étude se focalise sur les fondamentaux du marché de la
télémédecine et décrit en détail l’environnement du marché, aboutissant à une
analyse de marché « FFOM » (Forces, Faiblesses, Opportunités, Menaces).

Market study on telemedicine
Final Report
17
De cette partie de l’analyse de marché il apparait que l’adoption des technologies de
l’information en Europe est le principal accélérateur pour le déploiement de solutions de
télémédecine. Le potentiel du marché pour la télémédecine est très important et devrait
croître à un taux de croissance annuel composé de 14% dans les années à venir. Le
marché du « well-being » en particulier, grâce aux technologies digitales (dispositifs
portables et applications mobiles principalement), croît particulièrement rapidement.
Bien qu’il apparaisse que la demande pour des solutions de télémédecine dépasse
l’offre, cette observation est à considérer avec précaution, car de nombreuses initiatives
et solutions sont disponibles sur le marché, mais les hôpitaux et les cliniques n’ont pas
toujours les ressources financières pour adopter les technologies de pointe qui permettent
le déploiement des services de télémédecine.
Les entreprises de télécommunication, les fabricants d’électronique et d’outils TIC
(Technologies de l’Information et de la Communication), les fabricants de dispositifs, les
entreprises de l’industrie pharmaceutique et les « start-ups » ont été identifiés comme les
acteurs clés de la chaîne de valeur du marché de la télémédecine.
Dans un troisième temps, les obstacles à l’adoption de la télémédecine sont
identifiés dans l’une des sept catégories suivantes : culturel ; réglementaire et politique ;
sécurité sociale ; industriel et technique ; connaissances ; financier ; et lié au marché.
Sur la base de la revue documentaire, des obstacles ont été identifiés dans tous les
pays européens, mais sans les affecter de la même manière. C’est pourquoi il est
difficile de quantifier comment l’impact des obstacles varie selon le pays. De plus, la
télémédecine étant un marché avec de multiples parties prenantes, les obstacles
impactent les acteurs en présence différemment dans chaque pays considéré.
Les décideurs devraient être attentifs à ces obstacles et mettre en place des actions pour
les dépasser, notamment concernant : le conservatisme ou la résistance à l’adoption de
nouveaux processus médicaux, le manque d’interoperabilité entre la technologie et les
procédures des professionnels de santé, la législation (protection des données), le
manque de soutien financiers/d’incitations financières.
Il est important de noter que l’adoption de solutions de télémédecine à travers les
systèmes de santé nationaux ne sera fructueuse que si les institutions clés de la
communauté médicale, telles que les cliniques et les hôpitaux emblématiques, mettent en
place de nouveaux partenariats. Ces institutions ne seront encouragées à le faire que si
des décideurs nationaux permettent aux systèmes de santé de prendre en charge
correctement l’utilisation de cette technologie, ce qui signifie développer des
programmes de remboursement pour l’utilisation de la télémédecine. Au-delà de ce
point, il est important de souligner qu’aujourd’hui seuls les modèles en direct avec les
consommateurs ont du succès, car les acteurs institutionnels ne peuvent assurer une
prise en charge financière ou ne sont pas toujours remboursés pour les produits et
services de télémédecine.
Malgré les obstacles décrits ci-dessus, il convient de noter que dans de nombreux
domaines les initiatives nationales et de l’UE ont eu un impact très positif sur l’adoption
de la télémédecine, comme avec les « e-prescriptions » (prescriptions électroniques) par
exemple, ou le dossier du patient.
Enfin, cette étude présente une évaluation économique avec l’objectif de mesurer les
bénéfices potentiels du futur déploiement d’outils et de services de télémédecine à
travers l’UE. Cette évaluation repose sur une analyse elle-même basée sur des scénarios,

Market study on telemedicine
Final Report
18
qui utilise un modèle de décision économique. Les paramètres utilisés dans ce modèle
sont tirés de recherches scientifiques complétées par des données statistiques sur les
pathologies.
La première étape consiste à examiner des bases de données de recherche afin de
mettre en évidence des preuves du rapport coût-efficacité de la télémédecine dans
les essais cliniques. Les principales conclusions de cette analyse suggèrent que la
télémédecine serait efficace en termes de coût dans 73,3% des cas couverts par la revue
documentaire, tandis que les effets négatifs comptent pour 5,6% des études
sélectionnées. Les 21,3% restants des études analysées présentent un effet neutre sur
l’utilisation de la télémédecine comme moyen pour réduire les coûts. Les paramètres
qui ont un impact fort sur le rapport coût-efficacité des solutions de télémédecine
rapportées par les études incluent : distance entre le patient et le professionnel de santé
le plus proche ; temps requis par consultation ; coût de la visite d’un docteur ; année(s)
de vie pondérée(s) par la qualité ; et taux de mortalité.
Dans une deuxième étape, ces paramètres de coût sont utilisés pour mesurer le rapport
coût-efficacité qui résulte d’un déploiement à grande échelle de la télémédecine
en Europe, en se basant sur différents niveaux d’adoption projetée. Dans cette dernière
partie, l’étude se penche sur deux scénarios. Dans le premier scénario, il est supposé que
18% des soins de santé, la consultation et le traitement principalement, ont lieu avec
l’utilisation de la télémédecine. Le second scénario examine l’impact d’une hausse du
niveau d’adoption de 5% à 23%. Dans les deux scénarios, les coûts et bénéfices de la
télémédecine sont comparés au parcours traditionnel du patient en face-à-face pour
estimer l’effet d’un déploiement plus grand de la télémédecine. Il apparait alors que plus
la part de télémédecine est importante, plus le déploiement à grande échelle
devient efficace en termes de rapport coût-efficacité. Une part croissante de la
télémédecine réduit le coût total du parcours du patient, le temps de consultation total, la
distance totale parcourue et les taux de mortalité, et augmente le nombre d’années de
vie pondérées par la qualité.
Cependant, il s’agit seulement d’une première évaluation à l’échelle de l’Europe. Les
décideurs politiques doivent investir pour obtenir plus de preuves scientifiques de
l’efficacité de la télémédecine en finançant et en pilotant des expériences à grande échelle
pour mesurer l’impact d’un déploiement de grande ampleur. Sensibiliser (patients,
docteurs), soutenir l’intégration entre les différentes parties prenantes et faciliter le
remboursement sont autant de considérations supplémentaires pour accélérer l’adoption
et l’obtention des bénéfices résultant de l’utilisation de la télémédecine.

Market study on telemedicine
Final Report
19

Market study on telemedicine
Final Report
20
INTRODUCTION
Background
Telemedicine has a long history, as indicated in the Figure below. It started in ancient
times, but evolved in the 19
th
century with the invention of electricity and radio, and in
the 20
th
century with the development of television and the Internet.
Figure 1: A short history of telemedicine
In recent years, technological development enabling data analytics, artificial intelligence
and the healthcare Internet of things has disrupted traditional medical operations and
transformed healthcare provision. The increase in connected wearables and health-related
applications makes it possible to deploy telemedicine solutions on a wide scale. In 2016,
79% of EU residents between 16 and 74 years old accessed the Internet using a mobile
phone or smartphone.
1
In the near future, robots will be able to perform surgery
autonomously or driven by surgeons remotely.
The use of telemedicine is driven both by consumers, who seek to take advantage of
technologies that can improve their health and quality of life, and by healthcare systems,
1
http://ec.europa.eu/eurostat/documents/2995521/7771139/9-20122016-BP-EN.pdf

Market study on telemedicine
Final Report
21
which are interested in providing quality services with a reduced budget. The need for
services is increasing due to a) the economic development that enables counties and
individuals to buy better healthcare services, and b) the increase in the number of
patients with chronic diseases as the post-war baby boom generation ages.
In addition, the physical geography of Europe, with many islands and remote areas,
motivates the wide deployment of telemedicine. Several pilot projects have taken place in
the last few decades and have recorded positive results in terms of both improvements in
health and cost-efficiency. In 2014, five main use cases were reported in EU Member
States:
2
Teleradiology – the remote assessment of X-ray images, including peer review;
Teledermatology services, providing advice and second opinions both to physicians
and directly to citizens/patients (based on images of their skin problems);
Telestroke services (teleneurology), enabling early stroke treatment (thrombolysis);
Telemonitoring for diabetes (with coaching support), improving lifestyle and
conditions;
Telemonitoring for chronic heart failure as a prototype for intensified patient care.
2
Widespread Deployment of Telemedicine Services in Europe”, report of the eHealth Stakeholder Group on
implementing the Digital Agenda for Europe, Key Action 13/2 (‘Telemedicine’), version 1.0 final (12 March 2014)

Market study on telemedicine
Final Report
22
Figure 2: Pilot telemedicine projects

Market study on telemedicine
Final Report
23
Recent technological advances have occurred to the extent that Healthcare 4.0 may be
possible in the near future by using big health data analytics and artificial intelligence.
3
Today, fast Internet connections are widespread, through both fibre-optic and mobile
networks (4G/5G), allowing for synchronous, uninterrupted video streaming (which is
necessary in many telemedicine applications). All smartphones sold by major market
players are equipped with a free application that can monitor the user’s physical activity.
At the same time, people may have already purchased smartwatches and smartphones
with applications that can monitor and feed data to a medical professional. Connected
wearables are the infrastructure that can deploy telemedicine for common chronic
diseases such as high blood pressure. Of course, telemedicine can be practised today in
many ways, using specific hardware and applications for C2B (patient to doctor) and B2B
solutions (health professional to doctor/clinic/hospital). One innovative example is the use
of drones for emergencies such as heart attacks, which could dramatically increase
survival rates as patients can be reached more quickly than by ambulance.
An emerging trend that could also form a significant part of the telemedicine market is
electronic visits to doctors. In 2015 in the US, 800,000 out of 930 million doctors’ visits
were e-visits.
4
This is less than 1% of all doctors’ visits that year in the US, while the
American Medical Association states that 75% of all doctors’ visits are either unnecessary
or could be handled via telemedicine. In Europe, some telemedicine services, such as
teleconsultation, are supported by start-up companies that allow patients to see a doctor
online.
5
However, the deployment of telemedicine to the whole population of a country depends
on the country’s level of digitalisation, including the digital skills of patients and health
professionals, as well as the legislation governing the sharing and processing of health
data. While teleconsultation can be easily deployed with a small investment (for example
in France each doctor will get an up to 525 Euros support for the necessary software
solutions), other telemedicine service require a significant initial investment. Thus, the
cost-effectiveness and the return on investment need to be assessed in the longer term
based on the current state financial and market conditions. Finally, risks related to
healthcare data privacy breaches when exposing more data in networks and online
platforms need to be taken into account and telemedicine application should be equipped
or supported by strong encryption solutions. Such challenges are already being faced by
countries inside and outside Europe
6
.
Objectives of the study
The purpose of this study is to provide a full analysis of the market for telemedicine
applications and solutions based on the current conditions. The data collected, the
analysis and the conclusions will serve to inform and shape the Commission’s policy on
3
http://www.kmgus.com/blogs/healthit/index.php/2016/12/healthcare-4-0-the-future-of-healthcare
4
https://medium.com/@guidohegener/telemedicine-in-europe-battle-mode-on-b6ff4076ba5c
5
For an example, see this UK-based solution: https://www.pushdoctor.co.uk/
6
http://www.dsih.fr/article/3025/rendez-vous-medicaux-en-ligne-et-donnees-personnelles-le-scandale-
australien.html
http://www.dsih.fr/article/3037/singapour-l-infection-d-un-ordinateur-permet-le-vol-des-donnees-d-1-5-million-
de-patients.html

Market study on telemedicine
Final Report
24
telemedicine. In detail, the study is divided into four main tasks with the corresponding
number of work packages:
1. Mapping of existing solutions and relevant technical standards and/or guidelines;
2. Analysis of the market for such solutions, both in general and with regard to
specific sub-areas
3. Mapping exercise of barriers to the wider implementation of telemedicine, as well
as potential EU-wide approaches or solutions;
4. Most-effectiveness analysis of existing solutions and of potential wide-scale
deployment.

Market study on telemedicine
Final Report
25
KEY TERMS AND CONCEPTS
eHealth and mHealth
The World Health Organisation (WHO) defines eHealth
7
as the use of information and
communication technologies (ICT) for health. The terms ‘eHealth’ (electronic health) and
‘mHealth’ (mobile health) have been used in recent years to describe the provision of
health services using the Internet and mobile devices, respectively.
Telemedicine
Telemedicine is the provision of healthcare services where traditional face-to-face patient
- doctor interaction (or doctor - doctor) is replaced by over-distance interaction through
use of ICT. Several other definitions of telemedicine exist. Shaw
8
defines it as the use of
telecommunications technology for medical diagnostic, monitoring, and therapeutic
purposes when distance separates the users. The WHO has adopted the following
description:
9
the delivery of health care services, where distance is a critical factor, by all
health care professionals using information and communication technologies for the
exchange of valid information for diagnosis, treatment and prevention of disease and
injuries, research and evaluation, and for the continuing education of health care
providers, all in the interests of advancing the health of individuals and their
communities.
Telehealth
According to the WHO
10
, telehealth involves the use of telecommunications and virtual
technology to deliver health care outside of traditional health-care facilities, [for example]
a virtual home health care, where patients such as the chronically ill or the elderly may
receive guidance in certain procedures while remaining at home. Telehealth has also
made it easier for health care workers in remote field settings to obtain guidance from
professionals elsewhere in diagnosis, care and referral of patients. Similarly, Shaw
7
defines telehealth as the use of electronic information and telecommunications
technologies to support long-distance clinical health care, patient and professional health-
related education, public health, and health administration. Miller
11
suggests that
telehealth refers to both clinical and non-clinical applications in the way of
education, administration, and research while telemedicine is often reserved for
clinical, patient care applications.
There is a vast amount of literature with many definitions of the terms discusses above.
We can conclude that telehealth is a more generic term that refers to health-related
procedures, while telemedicine refers more specifically to treating people from distance.
eHealth and mHealth are terms that are as generic as telehealth in terms of health
7
http://www.who.int/ehealth/about/en/
8
Shaw, D. K. (2009). Overview of Telehealth and Its Application to Cardiopulmonary Physical Therapy.
Cardiopulmonary Physical Therapy Journal, 20(2), 13-18
9
http://www.who.int/goe/publications/goe_telemedicine_2010.pdf
10
http://www.who.int/sustainable-development/health-sector/strategies/telehealth/en/
11
Miller, E.A. (2007). Solving the disjuncture between research and practice: Telehealth trends in the 21st
century. Health Policy 82,133-141

Market study on telemedicine
Final Report
26
services, but specific to the technologies used in delivering these services from distance:
the Internet and mobile devices respectively. To make the latter explanation clearer, in
the case of telehealth, we may have patient-doctor interactions without Internet or
mobile devices.

Market study on telemedicine
Final Report
27
1. MAPPING AND CATEGORISATION
Key takeaways
Telemonitoring and prevention are the main types of intervention for telemedicine
solutions.
Telemedicine’s focus is on primary care, cardiovascular diseases (CVDs), chronic
obstructive pulmonary diseases (COPDs) and diabetes.
A great number of solutions target well-being and self-care, especially mobile health
applications.
Standards and guidelines mostly address technical requirements.
The aim of this chapter is to provide an analysis of the existing telemedicine solutions and
standards/guidelines through a systematic literature review (publications and reports).
The latter enabled us to highlight the main trends and characteristics of the telemedicine
solutions and standards. Another aim of this chapter is to put the EU/EEA status of
telemedicine into an international perspective, comparing it to the United States, Canada
and Japan.
1.1. Analysis of telemedicine solutions
Telemedicine solutions can be described as products and services designed to utilise
technology to improve and coordinate patient care, address growing health costs and
confront the long-term burden of disease. This sector is revolutionising the healthcare
industry through numerous applications in the fields of healthcare prevention and patient
management and monitoring. The tools and solutions that have emerged in recent years
are at the core of improved healthcare services provided by public and private
organisations. These digital tools increase healthcare delivery efficiency, enable patients
to be monitored remotely, improve access to electronic health information, enhance the
quality of healthcare services, and reduce costs.
The uptake of telemedicine solutions has enabled healthcare service providers to improve
patient-management processes through remote monitoring and follow-up, ensure the
continuity of access to day-to-day care, and create a wider information base for clinical
decision-making. Therefore, the uptake and wider implementation of these solutions
across healthcare providers has the potential to bring positive effects in key healthcare
fields such as chronic disease management.
These solutions comprise applications and tools that enhance the provision of healthcare
services on a remote and distant basis. This characteristic addresses the need to ensure
access to healthcare services for patients located at a distance from hospitals and clinics,
and eases the process of prevention, patient management, follow-up and monitoring. This
translates into concrete clinical health services that include teleconsultation,
telemonitoring, tele-education, telecare and telesurgery, amongst others. These systems
enable one or more patient disorders to be managed properly. For instance, patients
suffering from heart and blood pressure ailments can be monitored on a daily basis,
making treatment easier and more effective.

Market study on telemedicine
Final Report
28
1.1.1. Technical considerations
This section refers to the discussion of the technical features of telemedicine solutions.
Regarding the types of solutions, it appears from the analysis that products and platforms
were prevailing. Regarding the technical type, most of the solutions are medical devices
or include telemedicine support software. A specific section is dedicated to mobile
applications.
1.1.1.1. Types of solutions
The trend that emerges from our research is that most telemedicine solutions are
products and/or platforms. A telemedicine product is rarely marketed alone. Usually,
companies provide a platform (or an application) on which the data is shared. Then, the
data is stored in a database ready to be analysed and interpreted by a doctor, by another
healthcare professional or by software. This product-platform (-database)
combination is widespread in telemonitoring solutions. The Figure below provides an
illustration of data collection and sharing by the IT element of the telemedicine solution.
Figure 3: Data storage and sharing in tele monitoring software
For instance, the LifeWatch MCT 3 Lead is a mobile cardiac telemetry (MCT) product that
detects, records and wirelessly transmits asymptomatic and symptomatic arrhythmia to
clinicians for analysis. The four wearable cardiac electrodes are connected to a
smartphone via Bluetooth. If arrhythmia is detected, the smartphone automatically sends
the data to a monitoring centre for review and notifies a doctor if required.
Telemedicine services are often related to teleconsultations, telediagnoses or 24/7 call
centres. For example, the Swedish company Kry provides online video consultation with a
general practitioner holding a Swedish doctor’s license. The patient books an appointment
through the app, and then the doctor will call him/her, give him/her a 15 min consultation

Market study on telemedicine
Final Report
29
and send him/her a prescription, if needed. TeleRadiology Solutions provides radiology
interpretation through teleradiology services (e.g. CT, MRI, X-ray, ultrasound, nuclear
medicine, echocardiograms) to over 150 hospitals in 20 countries.
Databases are closely linked to platforms as well. They are useful tools for storing
medical information, especially electronic health records (EHRs). The Andalusian
eHealth Strategy & System DIRAYA in Spain, is a unified EHR system. It integrates
patients’ health information and intervention details in primary care, emergency services,
mental health services and specialist outpatient care.
Other ICT tools are quite marginal and correspond to solutions that either include other
technology or cannot fit into the given categories. For example, KineQuantum is a French
start-up that aims at projecting users/patients (undergoing physiotherapy) in 3D and
virtual-reality games. The idea is to have them perform exercises and specific movements
to measure and visualise their progress.
Applications, especially those designed for mobile devices, are much more numerous than
it appears in the mapping. Given the existence of hundreds of thousands of mHealth
applications, these are further discussed below.
1.1.1.2. Technical type
Figure 4: Technical type - data architecture in telemedicine solutions
The selections proposed in “Technical type” represent subsets of the selections proposed
in “Type of solution”. For instance, a “medical device” or “wearable device” corresponds
to a “product”, while a “mobile health app” refers to an “application”. Therefore,
conclusions can be drawn for the “Technical type” that are similar to those drawn for
“Type of solution” in the previous section.
A product-platform solution corresponds to a piece of telemedicine support software
integrated in a medical or wearable device. However, a telemedicine support software has
a wider scope, since it also encompasses services and databases. Behind almost every
telemedicine solution, there is a specific piece of software running because the latter

Market study on telemedicine
Final Report
30
is the fundamental technology that connects patients to healthcare professionals. This is
why these solutions account for most of the solutions in the mapping.
Figure 5: Telemedicine support software at the core of the solutions
Telemedicine products can be classified into two types: medical and wearable devices.
These two types often overlap because medical devices can be wearable devices and vice
versa. The WHO’s definition
12
for a medical device is broad.
Yet, a distinction has been made between wearable devices used mostly for personal
purposes (well-being, sport, fitness, etc.) and medical devices used in a medical
framework (i.e. in relations with a healthcare professional). For instance, the Polar Pro
strap developed by Polar Electro (Finland) is a soft textile strap with improved electrodes,
which measures the patient’s heart rate accurately. We considered this product wearable
but not a medical device. On the contrary, Biotronik Arrhythmia Monitoring (Biotronik,
Germany) is considered a wearable medical device for the purpose of this study, since it
allows healthcare professionals to review and monitor patients’ heart-rate data.
12
Medical device means any instrument, apparatus, implement, machine, appliance, implant, reagent for in
vitro use, software, material or other similar or related article, intended by the manufacturer to be used, alone
or in combination, for human beings, for one or more of the specific medical purpose(s) of:
• diagnosis, prevention, monitoring, treatment or alleviation of disease,
• diagnosis, monitoring, treatment, alleviation of or compensation for an injury,
• investigation, replacement, modification, or support of the anatomy or of a physiological process,
• supporting or sustaining life,
• control of conception,
• disinfection of medical devices
• providing information by means of in vitro examination of specimens derived from the human body;
and does not achieve its primary intended action by pharmacological, immunological or metabolic means, in or
on the human body, but which may be assisted in its intended function by such means.

Market study on telemedicine
Final Report
31
Big data/AI/Robotics are less common technical types of telemedicine solutions. The
combination of Artificial Intelligence (AI) and robotics might lead to new approaches in
surgery for instance. Up to now, Da Vinci's EndoWrist® is in fact only an improved
surgical procedure. The instrument bends and rotates far more than by conventional
laparoscopy but it’s still the surgeon who performs the medical act. Zebra Medical Vision
has created AI algorithms to read medical scans and detect anything untoward before
humans can.
1.1.1.3. Mobile heath applications – mHealth
The WHO’s definition of mHealth is also very broad: “medical and public health practice
supported by mobile devices such as mobile phones, patient monitoring devices, personal
digital assistants (PDAs) and other wireless devices”.
Worldwide – market size and growth
13
According to a study by Research 2 Guidance, in 2017 there were 325,000 mobile health
apps and 84,000 mHealth app publishers
14
in the five major app stores (Google Play,
Apple, Microsoft Windows Phone, Amazon, and Blackberry). Healthcare mobile app
development is one of the fastest-growing areas with a tremendous 32.5% CAGR
15
(41%
expected for 2015-2020), and reached €17.64bn in market revenues at the end of 2017.
Europe accounts for 30% of the market
16
(28% for the US). The global market is
predicted to reach €38.64bn by 2020
17
.
Europe is the fastest-growing segment in this market, with a CAGR of 61.6%.
18
13
N.B: the data available on mobile health apps includes both pure medical applications (used in medical
treatment) and applications related to self-care, well-being or lifestyle
14
Source: https://research2guidance.com/84000-health-app-publishers-in-2017/
15
Compound Annual Growth Rate (CAGR) = (Ending Value/Beginning Value)
(1/# of years)
- 1
16
GSMA and PwC, “Touching lives through mobile health - Assessment of the global market opportunity”,
February 2012
17
Source: https://www.statista.com/statistics/387867/value-of-worldwide-digital-health-market-forecast-by-
segment. Statistics published in US$ converted to Euros with an exchange rate of 0.84 Euros per US$
18
Dr Cheryl Lee Barton, BCC Research, Mobile Health (mHealth) Technologies and Global Markets (HLC162A),
March 2014
6,72
11,76
17,64
23,52
31,08
38,64
2015 2016 2017* 2018* 2019* 2020*
Market study on telemedicine
Final Report
32
Figure 6: Mobile health market value (in billion Euros)
Source: Statista (2018)
The number of mHealth app downloads has also dramatically risen for the past four
years, from 1.7 billion in 2013 to 3.7 billion in 2017 (+2bn in absolute terms, or +118%).
Figure 7: Number of mHealth downloads worldwide (billions)
Source: Statista – Research2Guidance
Leading European countries for mHealth apps
In a survey conducted by Research2Guidance in 2015, 4,471 mHealth app publishers and
decision makers were asked to rank the top three countries in Europe in terms of
favourable market conditions for mHealth business. The UK and Germany are the leading
countries, with 55% and 41% (respectively) of the mHealth app publishers and decision
makers mentioning them in the top three. We notice a strong attractiveness towards
Scandinavian countries as well (Sweden 23%, Denmark 16% and Finland 15%).
1,7
2,3
3
3,2
3,7
0
0,5
1
1,5
2
2,5
3
3,5
4
2013 2014 2015 2016 2017

Market study on telemedicine
Final Report
33
Figure 8: Share of mHealth publishers by country
Source: Statista – Research2Guidance
Disease specifics
Almost 1 in 3 mobile health apps are dedicated to mental health. Mental health relates to
mental and psychological well-being (WHO). The available solutions are very diverse.
Example include breathing exercises for stress management (Breathe2Relax); alert
notifications to specific contacts for teenagers struggling with depression or bullying
(Code Blue); and cognitive behavioural therapy (CBT) techniques with advice from real
experts (Lantern).
In the 2013 study conducted by IMS Health,
19
the categorisation of endocrine included
diabetes and metabolic syndrome, but in the 2015 study, these were categorised
separately. Diabetes and heart/circulatory diseases are the next most treated diseases by
mobile health applications: in 2015, 15% and 10% of disease-specific apps focused on
these two diseases respectively.
19
Statista, mHealth, November 2016
55%
41%
23% 23%
18%
16%
0%
10%
20%
30%
40%
50%
60%
United
Kingdom
Germany Sweden Netherlands Denmark France

Market study on telemedicine
Final Report
34
Figure 9: Distribution of disease-specific apps available worldwide in 2013 and 2015
Source: IMS Health
Health-context considerations
All the categories of mobile health apps (see Figure 10 below) are considered very
promising by app publishers in terms of market potential. Remote monitoring devices
increasingly use smartphone applications to store and monitor the data. Products are
regularly being developed that synchronise with smartphones, enabling patients to
monitor their conditions anywhere, anytime
20
,
21
. Over 70% of mHealth app market
players choose to publish their apps on both iOS and Android platforms.
Figure 10: Mobile health app categories that will offer the highest global market potential in the
next five years, as of 2016
20
Research2Guidance, “mHealth App Developer Economics 2016”, October 2016
21
European Commission, “COM(2014) 219 final GREEN PAPER on mobile Health (mHealth)”, April 2014
28%
14%
7%
6%
29%
15%
10%
7%
0% 5% 10% 15% 20% 25% 30% 35%
Mental health
Endocrine
Heart/circulatory
Musculoskeletal
2015 2013
32%
31%
30%
29%
28%
27%
26%
0% 5% 10% 15% 20% 25% 30% 35%
Remote monitoring
Diagnostic apps
Medical consultation management
Remote consultation
Patient health record
Fitness tracking
Nutrition
Share of respondents (2,600 publishers)

Market study on telemedicine
Final Report
35
Source: Research2Guidance
1.1.2. Geographical distribution
1.1.2.1. The EU leading countries
Three indicators have been selected in order to apprehend the geographical distribution
of telemedicine solutions, as indicated in the illustration below:
Figure 11: Indicators of the telemedicine market in Europe
Proportion of companies on the Integrated Personal Health/Care Services (IPHS)
market per country
The number of companies on the IPHS market in Europe provides an overview of how the
telemedicine solutions are distributed geographically. IPHS is a subcategory of
telemedicine with a similar scope. According to the EU-funded project Strategic I
SIMPHS 2) Technical Annex:
22
“Integrated Personal Health/Care Services address the
health and/or social care needs of individuals outside of care institutions and support the
work of care providers in an integrated fashion. IPHSs:
a) Can integrate assistance, remote monitoring of chronic diseases, wellness and
fitness;
b) Are produced as a result of integration of different institutional and information
systems. They are personal and possibly personalised in the way they gather,
process, and communicate data (for feed-back/action) and in terms of technological
components they can include”.
22
Baum P., Abadie F., “Market Developments – Remote Patient Monitoring and Treatment, Telecare,
Fitness/Wellness and mHealth”, JRC Scientific and Policy Reports, 2013.

Market study on telemedicine
Final Report
36
Figure 12: Proportion of IPHS companies per EU country
Source: SIMPHS 2 (2013)
The data referring to the IPHS companies on the market dates back to 2013 and takes
into account 64 European IPHS companies identified by the Joint Research Centre. The
proportional distribution of IPHS companies shows that Germany and the United Kingdom
are the two European leaders in providing telemedicine solutions (see Figure 12 above).
The same results emerge from our mapping: Germany and the United Kingdom are the
two European countries in which telemedicine solutions are mostly used. The latter
finding is expected give the population sizes of these countries.
Revenue distribution per country in the telehealth market
While – not surprisingly – Germany, France, the UK and Italy have a large proportion of
telehealth market revenue given that they are among the largest EU countries, it is also
interesting to note that if we aggregate the telehealth market revenues of Denmark,
Sweden, Norway and Finland, Scandinavia appears to be a dynamic region in the market
with revenues of over 129m euros. This is nearly 9% of total telehealth market revenues.
30%
19%
12%
8%
6%
5%
3%
17%
Germany
United Kingdom
Sweden
Netherlands
Ireland
Italy
France
Other

Market study on telemedicine
Final Report
37
Figure 13: Telehealth revenue distribution in European countries (2016)
Source: Statista (2016)
Revenue distribution per inhabitant in the telehealth market
Indeed, when the telehealth market revenues are divided by each country’s population,
then the Scandinavian countries become EU leaders. Their telehealth market revenues
per capita, especially in Denmark, exceed those of the United Kingdom and France. Of
course, the living and medical costs in these countries are much higher to eastern and
southern European countries.
22%
16%
15%
12%
8%
5%
4%
3%
3%
14%
Germany
France
United Kingdom
Italy
Spain
Netherlands
Poland
Sweden
Belgium
Other

Market study on telemedicine
Final Report
38
Table 1: Telehealth market revenue per inhabitant in euros
Country
eHealth market
revenue per
inhabitant (€)
Denmark
6.22
Sweden
5.05
Netherlands
4.75
Germany
4.69
Austria
4.66
Finland
4.46
France
4.09
United Kingdom
3.92
Belgium
3.87
Italy
3.38
Ireland
3.21
EU-24 average
3.05
Spain
2.97
Slovenia
2.68
Portugal
2.42
Czech Republic
2.26
Slovakia
2.14
Lithuania
1.98
Latvia
1.89
Croatia
1.80
Estonia
1.69
Poland
1.62
Hungary
1.25
Bulgaria
1.11
Romania
1.04
Further to the magnitude of the revenue per individual, which provides insight on the
base of added value in the telemedicine sector per country and population, it is important
to observe the efforts and advancement of different countries from a different
angle. One way to illustrate such level of advancement of EU countries is to look at
other factors of development such as the level of acceptance by the population and
the speed of uptake of telemedicine solutions.
One indicator that can provide insight on the level of advancement of a country
concerning the uptake of telemedicine tools and services is the use of electronic
networks and infrastructure by general practitioners in order to transfer
prescriptions to pharmacists, enabling a telemedicine solution that can improve
patient management and follow-up.
In this specific domain, studies have shown that up to 2013, the top five EEA countries in
terms of e-prescriptions were Estonia, Denmark, Croatia, Sweden and Iceland with nearly
the full population of general practitioners using remote technologies for the transfer of
prescription to pharmacists in digital format.

Market study on telemedicine
Final Report
39
Figure 14 : Use of electronic networks for ePrescription (% of GPs)
Source: European Commission (2013). Benchmarking deployment of eHealth Among General Practitioners II.
The Commission study on the deployment of eHealth also indicates that the top five
countries where patient data exchange was the most accepted and diffused
amongst general practitioners were Denmark, the Netherlands, Estonia, Iceland and
Finland.
Figure 15 : Patient data exchange with healthcare providers (% of GPs)
Source: European Commission (2013). Benchmarking deployment of eHealth Among General Practitioners II.
The observations made above show that even though wide revenues per capita in the
sector of telemedicine can be made in northern and western European countries, it is in
northern and eastern European countries where the adoption of telemedicine services and
tools amongst health professionals and hence users is the fastest.
The outlook of the wide deployment of such tools and services across Europe depends not
only on the size of national markets, but also on the speed of adoption by health
professionals and by end users.
100,0
99,6
98,8
97,3
96,2
94,0
85,7
82,3
59,8
52,7
28,5
20,6
17,8
16,0
15,4
11,0
9,2
8,1
7,8
6,0
5,0
4,9
4,6
4,4
3,5
3,1
2,2
0,6
0,5
0,0
0,0
10,0
20,0
30,0
40,0
50,0
60,0
70,0
80,0
90,0
100,0
Percent of GPs
91,8
76,2
72,0
67,9
66,8
65,7
63,6
56,0
52,8
42,9
39,4
39,4
37,0
31,2
28,8
24,0
23,9
23,4
18,0
17,8
16,9
15,9
13,0
12,2
12,0
11,0
10,8
8,7
7,5
5,4
0,0
10,0
20,0
30,0
40,0
50,0
60,0
70,0
80,0
90,0
100,0
Percent of GPs

Market study on telemedicine
Final Report
40
1.1.2.2. Scope of solutions by EU-based companies
Most solutions developed by EU companies have a national or regional market.
Indeed, European market players first try to conquer national (or at least, regional)
markets before taking the leap internationally.
Difficulties in entering markets in other EU countries or countries outside the EU are
linked to regulatory fragmentation (different rules applicable to telemedicine in
different countries) as well as restrictions of the Social Security schemes when it
comes to their proposition to the patient. Overall, the interoperability of a telemedicine
product/solution will be a crucial challenge in the next few years so that this
product/solution can enter the global markets.
The European Commission, through its eHealth Action Plan 2012-2020, Digital Single
Market policy and related initiatives
23
is endeavouring to strengthen the
interoperability of telehealth systems between Member States and thereby cross-
border use of telemedicine solutions.
1.1.3. Healthcare-context characteristics
Another result of our mapping is the identification of recurrent health-context
characteristics. The major findings are:
Prevention and telemonitoring are the more common usages for telemedicine
solutions.
Telemedicine solutions aim mostly at providing primary and home-based care.
The main market segments are solutions for patient-doctor interaction, solutions for
healthcare professionals’ collaboration, and self-care solutions.
23
For instance the recent communication on enabling the digital transformation of health and care in the Digital
Single Market, ttps://ec.europa.eu/health/sites/health/files/ehealth/docs/com2018_233_en.pdf

Market study on telemedicine
Final Report
41
Figure 16: The main types of health context
1.1.3.1. Health-context considerations
The study SIMPHS 3 (2015), conducted by the Joint Research Centre, exhibits the
distribution of telemedicine solutions by type of intervention provided (from a sample of
86 solutions). The different types of intervention highlighted do not include
telemonitoring. The graph below displays the distribution of telemedicine solutions
among these types.
Figure 17: Type of intervention for telemedicine solutions
Source: SIMPHS 3 (2015)
Prevention; 41%
Treatment; 23%
Prevention and
treament; 17%
Other; 19%

Market study on telemedicine
Final Report
42
Treatment for a disease is a type of intervention provided by 40% (23+17) of the
telemedicine solutions. From our mapping, there are slightly fewer solutions that
administer treatment. We understand medical treatment as the management and care of
a patient to combat disease or disorder. Technologies able to cure or treat a disease
directly seem less prevalent than they appear.
However, prevention is the dominant type of intervention covered by telemedicine,
present in 58% of the solutions. We assume that telemonitoring has been included under
prevention in this study, since telemonitoring contains reviews and follow-ups by
professionals to reduce the occurrence of complications. Remote patient monitoring
seems to be the most widespread telemedicine solution, as the existing technologies
enable this medical practice to be implemented effectively.
24
Voluntary (or unspecified) usage largely outweighs mandatory usage in our mapping. This
is due to our methodology approach, which consists of considering a solution mandatory
only when it is clearly specified. Another hypothesis is that conditions for mandatorily
adopting telemedicine solutions in a healthcare programme have not yet been considered.
These barriers from adoption are treated in Work Package 3.
1.1.3.2. Level of care usage
Remote patient-monitoring devices are meant to increase residential and home based
care. Hospitals use these solutions to substantially lower costs and risks related to
hospitalisation. Indeed, by implementing suitable follow-up care and care management
of patients at home, hospitals can prevent unnecessary readmissions.
Many of the solutions also aim at providing primary care to patients. Primary care
providers such as general practitioners (GPs) can take a lot of time following up with
patients coming into their office. Telemedicine offers appropriate means to save time for
both practitioners and patients without compromising on care efficiency. Using
the Telea Digital Home Platform developed by Sergas (Spain), a single healthcare
professional can monitor up to 50 patients through videoconferencing, electronic health
records, custom notifications, etc.
24
“Strategic Intelligence Monitor on Personal Health Systems, Phase 3”, 2015

Market study on telemedicine
Final Report
43
Figure 18: Different levels of care usage
Telemedicine solutions for specialist care providers are also becoming an integral part of
healthcare delivery. They allow for patients with chronic conditions to be better managed,
thanks partly to the remote monitoring devices. This also affects the patients’ care
pathway, as primary care doctors have easy access to specialists. This way, the specialist
can make an immediate diagnosis and the primary care provider can start a treatment
plan rather than sending the patient to the specialist.
For instance, Dermtest (Estonia) is a software platform connecting general practitioners
with dermatologists, to provide an early skin-cancer detection service to patients at their
local general practitioner’s office.
1.1.3.3. Stakeholders
Our analysis shows that the market is mainly divided into two segments: a) solutions
between healthcare professionals and patients (B2C), and b) devices for self-care. An
illustration of the interaction between doctors, patients and health professionals is present
in Figure 19 below.

Market study on telemedicine
Final Report
44
Figure 19: Interactions between stakeholders
Currently, one out of every two telemedicine solution targets self-care. In comparison,
solutions for professionals are lagging behind. These findings are in line with our market
research for 2017. There are only a few solutions involving doctor-to-doctor or doctor-to-
healthcare-professional interactions. It seems that the health market is characterised by
slow adoption rates for solutions targeting collaboration among professionals
(B2B). It merits note that the EU supports cross-border collaboration between health
professionals. One such initiative by the EU is the eHealth Digital Service
Infrastructure
25
(eHDSI or eHealth DSI), which is the initial deployment and operation
of services for cross-border health data exchange under the Connecting Europe Facility
(CEF). Another is the European Reference Networks (ERNs) – virtual networks involving
healthcare providers across Europe. Indeed, ERNs aim to tackle complex or rare diseases
and conditions that require highly specialised treatment and a concentration of knowledge
and resources. There are 24 ERNS involving 25 European countries included Norway, over
300 hospitals with over 900 healthcare units and covering all major disease groups.
26
Solutions for the interaction between patients and health care professionals utilise
technology that provides more efficient care delivery. Such technology operates as a
support mechanism and does not completely disturb the traditional doctor-patient
relationship. However, devices for self-care do challenge this long-established
relationship. Patients equipped with such devices may be able to take care of themselves,
regardless of any doctor’s intervention. Solutions for self-care might run counter to
telemedicine, which is defined as the provision of healthcare services through use of ICT
in situations where the health professional and the patient (or two health professionals)
are not in the same location. Yet, all solutions relating to self-care ended up in the
“Patient to doctor” category, as we consider that doctors can supervise self-care
treatments.
25
https://ec.europa.eu/cefdigital/wiki/display/EHOPERATIONS/eHDSI+Mission
26
https://webgate.ec.europa.eu/ern/

Market study on telemedicine
Final Report
45
Figure 20: Solutions by market segment
Source: SIMPHS 3 (2015)
1.1.3.4. Specific medical aspects
The majority of telemedicine solutions mainly target primary care highlighting its
importance. There are also solutions for specific medical specialties but these are more
limited in scope. Most solutions with a medical specialty concern heart failure, diabetes
and COPD, which concern a significant part of patients with chronic diseases.
Medical specialties
From our research, telecardiology, telepulmomology and teleendocrinology are the
three medical specialties that account for most of the telemedicine solutions. The
underlying hypothesis is that these specialties are particularly well suited to a
technological and virtual-care environment. They are fully exploiting the currently
available technologies to design devices and software tailored to patients’ needs.
The above three solutions are followed by teledermatology, teleoncology, teleneurology
and telemental health. Solutions in these specialties exist but are less widespread
because they usually require more advanced technologies. For instance, many watches
can measure heart rate and blood pressure, but no common devices exist to measure
neurological activity.
Besides, a large number of telemedicine solutions reported in our mapping do not refer to
any specific medical specialty. In particular, this observation concerns platforms that aim
at connecting patients to specialist doctors.
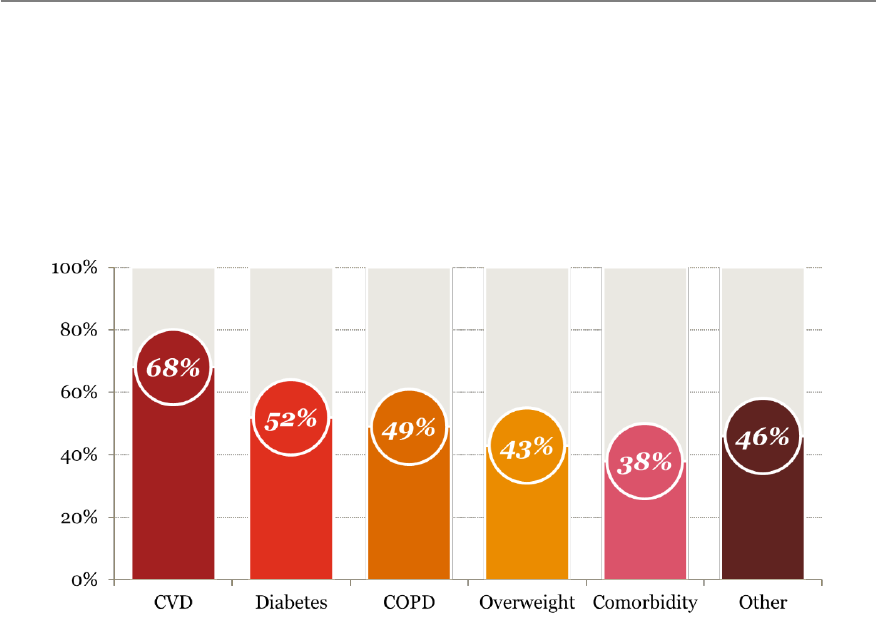
Market study on telemedicine
Final Report
46
Disease specifics
Unsurprisingly, the results for disease specifics corroborate those observed for medical
specialists.
The chart below displays how many European companies on the IPHS market address the
main diseases. In summary, cardiac conditions are addressed by 68% of the companies,
diabetes and COPD by about 50%.
Figure 21: Companies on the IPHS market – medical focus
Source: IPTS – SIMPHS 1 (2011)
Cardiovascular diseases, diabetes and chronic obstructive pulmonary disease are the
most treated by companies. This is consistent with the results for medical specialties,
where telecardiology, telepulmonology and teleendocrinology accounted for the most of
the telemedicine solutions.
1.1.4. Analysis of the status of telemedicine solutions
In this section, we focus on the timing of development of the telemedicine solutions.
These solutions are in various stages of maturity, from pilot phase to operational for more
than five years. Our analysis reveals that among the solutions studied as part of the
mapping exercise solutions operational for more than five years are slightly more
common.

Market study on telemedicine
Final Report
47
Figure 22: Levels of development of telemedicine solutions
Indeed, the situation as of 2013 shows that companies existing for more than five years
are dominating the market. Again, as IPHS is very close to telemedicine, the chart in
Figure 23 provides significant conclusions as regards as the status of telemedicine
solutions in 2013.
Figure 23: IPHS company distribution – years in business
Source: SIMPHS 1 (2011), SIMPHS 2 (2013)
It is apparent from our research that most solutions have been operational for more than
five years. This means that the first versions of these solutions should have been
launched before 2013, although they are likely to have evolved since then. On the
12%
22%
66%
< 2 years
2 - 5 years
> 5 years

Market study on telemedicine
Final Report
48
contrary, telemedicine companies keep upgrading their products, platforms and software
so that they can incorporate new attractive options and the latest technology
available. They also offer the same product at different quality levels (and thus, prices),
in order to target different segments of patients with various income levels.
Yet, new innovative solutions are increasingly gathering momentum in telemedicine
thanks to the multiplication of start-ups and the initiatives of hospitals, research centres
and universities. For instance, the University Hospital of North Norway (NST) and the
Austrian Institute of Technology (AIT) are developing their own blood glucose
measurement and management systems, which are currently in the pilot phase.
1.2. Analysis of standards and guidelines
Before presenting the results of the desk research on standards and guideless it is
necessary to distinguish these terms and provide definitions that will allow for a better
reading of this section. The definitions for the terms “standard” and “guideline” are
provided in Figure 24 below:
Figure 24: Definitions of standard and guideline
The current standards and guidelines identified have been classified in three ways: by
typology of issuing bodies (e.g. EU standardisation, medical association, national
standardisation), by geographical area (Europe, North America, international), and by
category (data-protection rules, human resources guidelines, technology and equipment
guidelines/standards, clinical guidelines, ethical guidelines, organisational guidelines, and
EHR guidelines).
1.2.1. Typology of issuing bodies
The supranational standardisation bodies account for most of the existing standards and
guidelines. Bodies such as the International Organization for Standardization (ISO) and
the ITU Telecommunication Standardization Sector (ITU-T) have published hundreds of
guidelines for providing telemedicine services. In the European Union, CEN is the
supranational body that issues the most standards and guidelines.
As already stated, few telemedicine standards and guidelines spring from national
standardisation bodies’ publications, since they work in close coordination with CEN.
However, national medical associations supplement these national bodies with some
additional standards and guidelines. They usually have a medical focus and provide
precise requirements for medical specialties (e.g. the Società Italiana di Radiologia Medica

Market study on telemedicine
Final Report
49
(SIRM) for teleradiology). In North America especially, the American Telemedicine
Association (ATA, in the US) and more specific associations, such as the Canadian
Association of Radiologists, also issue numerous documents and guides providing best
practices and requirements for various telemedicine specialties.
1.2.2. Geographical spread
Most of the existing telemedicine standards and guidelines are international in scope.
This is due to the sustained activity of supranational independent bodies that release
numerous reports and documents each year. Their publications are usually free to
access so that best practices spread all over the world. The International Organization
for Standardization (ISO) is responsible for most publications of international standards.
Organisations from the US and the EU also play a key role in the publication of
telemedicine standards. The primary goal is to standardise the best practices between
their states and Member States respectively.
For the EU, the principal standardisation body is the European Committee for
Standardization (CEN), which brings together the national standardisation bodies of 34
European countries. CEN is committed to developing and delivering European standards
in close cooperation with ISO. Other European bodies are the European Committee for
Electrotechnical Standardization (CENELEC) and the European Telecommunications
Standards Institute (ETSI), which are very involved in telemedicine.
At national level, action on standards and guidelines relies on national standardisation
bodies and medical associations. In the EU, national standardisation bodies aim at
coordinating standardisation in the country, in collaboration with CEN, to which they
belong. From our research, it appears that these national bodies are not as prolific as
international or supranational bodies. For each country, we found a few standards and
guidelines issued by national standardisation bodies. Our mapping shows that the UK’s
and France’s bodies seem to publish the most national telemedicine standards in the EU.
1.2.3. Guideline/standard category
Technology and equipment guidelines/standards are the most widespread telemedicine
standards. This result was quite predictable, since technology is at the core of
telemedicine practice. Some examples of technology standards and guidelines include:
the format and quality necessary for medical images in teleradiology; the performance
requirements for software and medical products; the broadcast capability; video coding
and decoding methods for moving pictures; and the clarification of the vocabulary specific
to the security of information systems. Other standards/guidelines specify general and
functional requirements for the use of the product/service.

Market study on telemedicine
Final Report
50
Figure 25: Typology of standards and guidelines
Data-protection rules are also quite common. They aim to control how personal data
can be used and ensure the patient’s rights regarding his/her information. For
instance, ISO/TS 17975 defines the set of frameworks of consent for the collection, use
and/or disclosure of personal health information by healthcare practitioners or
organisations. Another illustration is the Code of Conduct on privacy for mHealth apps
issued by the European Commission in 2016, which is currently under review after the
comments provided by the WP29
27
. It targets app developers and provides specific and
accessible guidance on how European data-protection legislation (the General Data
Protection Regulation - GDPR
28
) should be applied in relation to mHealth apps. At this
stage, mHealth apps need to comply with the GDPR.
Organisational guidelines outline the way in which business is to be conducted and
govern what is deemed acceptable workplace behaviour. The ISO 9001 standard is
based on a number of quality management principles, including a strong customer focus,
motivation and involvement from top management, process approach and continual
improvement. It is supposed to help ensure that customers get consistent, high-quality
products and services, which in turn brings many business benefits.
27
http://www.osborneclarke.com/insights/mhealth-apps-the-code-of-conduct-on-privacy-explained/
28
https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32016R0679&from=EN

Market study on telemedicine
Final Report
51
Clinical guidelines recommend how healthcare professionals should care for
people with specific conditions. They can cover any aspect of a condition and may
include recommendations about providing information and advice, prevention, diagnosis,
treatment and longer-term management. A Concise Guide for Telemedicine Practitioners
Human Factors: Quick Guide Eye Contact, issued by the American Telemedicine
Association (ATA), is summarises eye contact and its importance for telemedicine
practitioners delivering remote healthcare services. The guide covers the importance of
eye contact, eye-contact etiquette, tips for camera positions and viewing screens,
clinician positions, and other key tips for optimising healthcare provider-patient
interactions and relationships.
Human resources guidelines intend to inspire, educate and support board members,
managers and employees with regard to the fundamental role that HR
management policies and practices have in creating effective organisations.
Standards such as the International Code of Practice for Telehealth Services 2017
(Telehealth Quality Group) cover aspects including staff management, building a prepared
and competent team, and the qualifications and responsibilities of personnel in a
telemedicine environment. Nonetheless, human resources standards are rarer.
Ethical guidelines embrace a broad array of concepts. The American Medical Association
(AMA) provides recommendations regarding:
- Managing conflicts of interest: physicians should provide objective and accurate
information;
- Privacy and security: services must have appropriate protocols to protect the
security of patient information and prevent unauthorised access to such
information;
- Patient consent; and
- Standards of care: physicians should uphold the standards of professionalism
expected for in-person interactions and adhere to applicable law governing the
practice of telemedicine.
EHR guidelines are intended to aid healthcare providers and healthcare IT
implementers with in implementing an EHR system. The overall goal of ISO 13606
is to define a rigorous and stable information architecture for communicating part or all of
the electronic health record (EHR) of a single subject of care (patient). In addition to this
standard, ISO 18308 defines the set of requirements formulated to ensure that these
EHRs are faithful to the needs of healthcare delivery, are clinically valid and reliable, are
ethically sound, meet prevailing legal requirements, support good clinical practice, and
facilitate data analysis for a multitude of purposes.
Standards and guidelines aim to spread good practices and guarantee a certain level of
requirement in the use of telemedicine solutions. They also aim to ensure
interoperability between different devices, systems, organisations and
countries. To increase the adoption of telemedicine in healthcare, it is fundamental to
create conditions whereby solutions are compatible with other systems.
Thus, some institutions work to make sure that established standards are well
coordinated. For instance, Integrating the Healthcare Enterprise (IHE) promotes the
coordinated use of established standards such as DICOM and HL7 to address specific
clinical needs in support of optimal patient care.
29
IHE also tests the interoperability of
29
https://www.ihe.net/About_IHE/

Market study on telemedicine
Final Report
52
health information technology (HIT) systems. PCHAlliance, a non-profit organisation
formed by HIMSS, encourages the global adoption of the Continua Design Guidelines, an
open framework for user-friendly, interoperable health-data exchange in personal
connected health.
30
It is relevant to mention here the work of the eHealth Network as well, which is composed
of members coming from the 28 EU Member States and Norway (as an observer) and
holds biannual meetings
31
to discuss issues regarding patient access to electronic health
record information, interoperability and standardisation. The Network has developed
guidelines in relation to ePrescription and the Patient Summary:
- The Guidelines on ePrescriptions dataset for electronic exchange under Cross-
Border directive 2011/24/EU: Release 1
32
and the Guidelines on the electronic
exchange of health data under Cross-Border directive 2011/24/EU: Release 2
ePrescriptions and eDispensations
33
have been prepared and validated by the
eHealth Network in 2014 and 2016, respectively;
- Similarly, the eHealth Network first adopted the Guidelines on
minimum/nonexhaustive patient summary dataset for electronic exchange in
accordance with the Cross-Border Directive 2011/24/EU
34
in 2013. Three years
later (in 2016), after a revision, the eHealth Network adopted the Guideline on the
electronic exchange of health data under Cross-Border Directive 2011/24/EU:
Release 2 Patient Summary for unscheduled care
35
.
1.2.4. Medical specialties
From our research, the vast majority of standards and guidelines do not pertain to any
medical specialty. Recommendations formulated by bodies often lay down principles
and good practices about telemedicine in general, without focusing on any specific
discipline.
Telecare, telecardiology, teleradiology and teleendocrinology are the specialties most
targeted by standardisation bodies. Obviously, this relates to their level of development
and use: the more solutions, the more standards and guidelines.
30
http://www.pchalliance.org/personal-connected-health-alliance
31
https://ec.europa.eu/health/ehealth/events_en#anchor0
32
https://ec.europa.eu/health/sites/health/files/ehealth/docs/eprescription_guidelines_en.pdf
33
https://ec.europa.eu/health/sites/health/files/ehealth/docs/ev_20161121_co091_en.pdf
34
https://ec.europa.eu/health/sites/health/files/ehealth/docs/guidelines_patient_summary_en.pdf
35
https://ec.europa.eu/health/sites/health/files/ehealth/docs/ev_20161121_co10_en.pdf

Market study on telemedicine
Final Report
53
2. MARKET ANALYSIS
Key takeaways
The uptake of information technologies in Europe is the main accelerator for
telemedicine.
The market potential of telemedicine is strong. It is expected to grow at a compound
annual growth rate of 14% in the coming years.
The well-being market enabled by digital technologies (mobile applications, devices) is
rapidly growing as well.
Demand outpaces supply, but this should be read with care, as there are many
telemedicine initiatives but hospitals and clinics do no have the means to pay for the
technology.
Market players include: telecommunication companies, ICT tools and electronics
manufacturers, device manufacturers, pharmaceutical industry companies, and start-
ups.
US and Canada have outperformed the EU, whilst Japan has a lower number of users
of telemedicine.
Key objectives addressed by the uptake of the telemedicine market are the improvement
in patient management, treatment and care, coupled with a reduction in costs at both
individual and societal levels. Telemedicine itself is part of a larger framework of
connected healthcare, which takes the perspective of both institutional and individual
users of health services provided in a remote manner through data transmission and new
digital technologies.
Source: XERFI, 2017
Figure 26: Telemedicine in context, the connected healthcare framework

Market study on telemedicine
Final Report
54
2.1. Market fundamentals
Telemedicine covers a very wide area of products, services, procedures and techniques.
In essence, it designates all aspects relating to the progressive transformation of the
health sector due to the introduction of ICT, and relies on continuous investment in digital
infrastructure and digital skills in the healthcare industry. As part of a set of health
information technologies, telemedicine has gained more visibility amongst governments
and market players in recent years; these technologies play a key role in the European
Union’s digital strategy and have become one of the lead initiatives to create an
innovative Europe in a dynamic, knowledge-based economy.
36
Therefore, telemedicine
technologies require not only the use of information technology (including hardware,
software, telecoms and IT services), but also the leveraging of skilled human resources to
enable healthcare services to be delivered from distance or a remote location.
The principal telemedicine market drivers and trends during recent years have therefore
been an uptake and democratisation of information technologies, which have enabled the
remote transmission of information at ease, speed and marginal cost. These information
technologies have progressively defined a commercial ecosystem of health information
technologies, which is currently experiencing rapid growth globally.
37
According to market
figures provided by Statista, the global telemedicine market was valued at €16.3 billion in
2015, and is expected to reach more than €37 billion by 2021, with a CAGR of 14%
during that period. This dynamic sector therefore has the potential to drastically influence
the delivery of efficient patient care at a lower cost for healthcare markets worldwide.
Figure 27 : Global telemedicine market size from 2015 to 2021 (in billion Euros)
2.1.1 Description of the solutions/products that make up the
telemedicine market
As described in Chapter 1 of this report, telemedicine solutions include products and/or
platforms that collect and store patient information and data that can be treated
remotely by a doctor, healthcare professional or analyst to interpret it and enhance
patient management, treatment and care.
36
European Commission & Directorate General Information Society. (2009). Study on the Legal Framework for
Interoperable eHealth in Europe. European Commission, 1–128.
37
https://www.trade.gov/topmarkets/health-it.asp
16,29
18,18
20,7
23,85
27,45
31,95
37,08
2015 2016 2017 2018 2019 2020 2021
Billion Euros

Market study on telemedicine
Final Report
55
These solutions often have multiple components, including hardware and software in
embedded systems sold as one product.
38
The most prominent systems in telemedicine
embed mobile technologies to ensure continuous functionalities related to the storage
and transfer of administrative and medical patient data to health personnel and
administrations. Furthermore, telemedicine solutions are increasing in complexity every
day. The trend in newer generations of solutions is advancing the types of functionalities,
which now include real-time communication, adaptive scheduling, resource management,
multitasking, artificial intelligence and the transfer of data from sensor to destination.
The use of these systems over recent years has intensified the development of remote
patient management and monitoring. According to market estimation figures provided by
Statista, the market revenue from wearable devices and services has constantly grown
over the last five years from €13 billion to €23.1 billion and is expected to increase by an
additional €9 billion by 2020.
2.1.2 Geographical distribution of the market
Telemedicine – and to a wider extent information health technology – encompass a sector
that, as we described earlier, has experienced continuous incremental global growth in
the last few years. Further analysis of available market data indicates a similar pattern of
continuous growth, albeit at different pace, across several economies.
In Section 1.1.2, the geographical distribution of telemedicine solutions in the EU was
estimated by studying the distribution of integrated personal healthcare services across
EU Member States for which data is available. Based on this estimate, we have observed
a concentration of companies in countries with relatively high healthcare expenditure per
inhabitant. In economies beyond the EU28, this analysis indicates that the size of the
telemedicine market, in estimated users per inhabitant, is growing across countries
leading in digital technologies.
38
Global Intelligence Alliance. (n.d.). Embedded Systems for Telemedicine in Germany. Retrieved from
https://www.cbi.eu/sites/default/files/study/product-factsheet-embedded-systems-telemedicine-sweden-
finland-denmark-electronics-electrical-engineering-2014.pdf

Market study on telemedicine
Final Report
56
Figure 28: eHealth market outlook. Total number of users in the market (millions) by geographical
region.
Source: Statista estimates – trends include per population for three principal diseases (diabetes,
hypertension and heart failure) and World Bank data on total population
According to Statista’s “Digital Market Outlook”
39
, the “number of users” covers users in
three main telemedicine product categories:
Connected medical devices (“smart devices”
40
);
Digital tracking and monitoring applications for smartphones and/or tablets (“apps”);
and
Telemedical services for patients at risk (“telemedical services”). Hardware and
software solutions for healthcare professionals (e.g. medical equipment for hospitals
and doctors’ surgeries) are not included.
Further to this trend, total market revenues associated with the number of users follows
similar pattern. This analysis of the potential market size is estimated by calculating the
number of users and the total revenue for three principal chronic diseases (diabetes,
hypertension and heart failure), and is expressed in total users and total Euros per
thousand inhabitants. It shows that although the use of telemedicine solutions and
information health technologies is expected to follow a similar pattern across the main
39
Statista (2015). Digital Market Outlook. Available at https://www.statista.com/outlook/digital-markets
40
The “Smart Devices” segment covers medical devices (hardware) that are equipped with dedicated interfaces
or SIM cards that serve to transmit measurement data across a wireless connection (e.g. via mobile networks,
WiFi, Bluetooth, M2M technologies, NFC, BLE). The selection of suitable equipment is dependent on the
individual health status of the patient and the severity and presence of other conditions (e.g. a combination of
severe heart failure and cardiac arrhythmia).
0
2
4
6
8
10
12
14
16
18
20
2014 2015 2016 2017 2018 2019 2020
Number of users (per thousand inhabitants)
European Union Canada Japan Switzerland United States

Market study on telemedicine
Final Report
57
economies under review, the gap between generated revenues is currently increasing and
diverging, indicating an increased ability of other economies to obtain larger shares of
value creation.
Figure 29: eHealth market outlook. Per capita market revenues (Euros/1000 population) by
geographical region.
Source: Statista estimates. Trends include per population for three principal diseases (diabetes,
hypertension and heart failure) and World Bank data on total population
In terms of total volumes of revenues and users, the US eHealth market is much larger
Canada’s and Japan’s eHealth market.
However, it is important to notice that Japan presents total revenues of €448m in 2018 in
the eHealth market that can be compared to the total revenues in the eHealth market in
leading EU countries; indeed, these revenues are close to Germany’s €554m and are
higher than France’s €400m and the UK’s €366m
41
. On the contrary, the eHealth market
in Canada represents half the size of the market in the above EU countries and for 2018 it
is expected at the level of 165 million Euros.
41
Source: https://www.statista.com/statistics/515701/global-comparison-ehealth-revenue-digital-market-
outlook/
0
1.000
2.000
3.000
4.000
5.000
6.000
7.000
8.000
9.000
2014 2015 2016 2017 2018 2019 2020
Total revenue (Euros per thousand inhabitants)
Canada European Union Japan Switzerland United States

Market study on telemedicine
Final Report
58
2.1.3 Focus on three key global markets
We have dedicated this section to the development of telemedicine in the United States,
Canada and Japan. Overall, our main observations indicate that:
The United States is the pioneer country worldwide in telemedicine,
Canada is a forerunner in binding market players to comply with national telemedicine
standards and interoperability; it has established certification processes, and is
considered an early adopter of data protection legislation for eHealth,
Japan is lagging behind in relative terms.
USA
From a global comparison perspective, most eHealth revenue is generated in the United
States (€3,210m in 2018). Furthermore, revenue is expected to grow at an annual rate
(CAGR 2018-2020) of 14.2%, resulting in a market volume of EUR 4,187 million in 2020.
The market’s largest segment is heart failure, with a market volume of EUR 1,319 million
in 2018. In addition, investment deals in the eHealth sector are mainly observed in the
USA.
Among eHealth solutions for diabetes, hypertension and heart failure, products for heart-
failure patients generated the highest revenue in 2016, at €854 million. Heart disease is
the major cause of death in the United States, accounting for 23.4% of deaths in 2014.
Like in the EU, prevention and treatment are the prevailing types of intervention for
telemedicine solutions. Ambient assisted living (AAL) only includes devices to track the
user’s health data at home, and cannot therefore fall under the telemonitoring category.
In the same way as the distribution of intervention types in the EU countries, we assume
that other telemonitoring solutions are split between prevention and treatment.
Figure 30: Distribution of eHealth revenue across three key categories of utilisation in the United
States in 2016
Source: Statista (2017)
Prevention;
42%
Ambient
assisted living;
12%
Treatment; 44%

Market study on telemedicine
Final Report
59
In summary, the United States can be considered a pioneer and leading country in
embracing telemedicine. Furthermore, telemedicine in the US has conquered
healthcare at all levels.
42
The following examples support this finding.
1. Hospitals use telemedicine to provide their patients with specialist care (chronic
disease monitoring, follow-ups) to prevent unnecessary and costly readmissions.
2. Telemedicine is a way for employers to establish cheaper insurance plans for
their own employees. Nine out of ten employees can use telemedicine services.
3. Telemedicine also concerns urgent and primary care, as it raises customer
retention and saves costs in 24/7 services.
4. Doctors use telemedicine to avoid unnecessary, time-consuming visits to their
offices.
Canada
The adoption of telemedicine in Canada has fallen in the last few years in comparison to
other developed countries. However, the country endeavours to catch up with these
countries, now that conditions for countrywide implementation have been fulfilled.
Across Canada, the delivery of care via telemedicine continues to expand. Telemedicine
grew by 45.7% from 2012 to 2014. From 2010 to 2016, telemedicine delivery more than
doubled. Besides, the delivery of telemedicine services through means such as remote
monitoring or teleconsultation is a tremendous opportunity for Canada, since the country
suffers from an unequal geographical distribution between trained medical professionals
and the general population. The increasing popularity of telemedicine solutions might
provide the large rural population with easier access to healthcare.
43
Actually, according to Infoway, both the public and medical professionals are embracing
telemedicine. In 2015, more than one in five hospitals made remote monitoring services
available and more than 350,000 Canadians used medical devices that capture and
transmit data electronically to their healthcare provider for monitoring and support.
Canada has been a forerunner in binding market players to comply with national
telemedicine standards and interoperability, which can be considered a strength for the
national market environment. Furthermore, in 2013, Canada was the only country to
establish a certification process that targets market players’ products and services.
Amongst other things, the process included a number of usability requirements such as
service levels, technical-support responsiveness and financial viability. Finally, Canada is
an early adopter of data protection in the field of eHealth, imposing since 2013 a signed
obligation, such as a data-sharing agreement or a contract for data recipients. These
documents aim to legally bind market players to the rules protecting the privacy and
confidentiality of the data to which they have been approved access.
44
In addition, given the close proximity of the United States offers Canada the opportunity
of benefits from strong relationships between Canadian and US companies that may
include learning effects for market players.
42
Dr Andrew Lin, “2017: Telemedicine in the US and beyond”, April 2017.
43
International Trade Administration.
44
OECD, 2013.

Market study on telemedicine
Final Report
60
Japan
We have already highlighted that the Japanese eHealth market is sizeable in volume but
rather small in relative terms.
In Japan, the financial cost of providing ample healthcare services to the population is
unsustainable, especially in a slow-growth, developed/mature market. In 2014, the
Japanese Government spent €346bn on the provision of healthcare services; such an
expenditure has triggered the search for new ideas to curb healthcare expenditure. As a
result, private-sector healthcare providers in Japan (through improved home care and
community-based care, enhanced by mobile health and telehealth solutions) are expected
to acquire a bigger market share for treatment services, particularly for the elderly
population. This development could also help expand knowledge of the health of the
Japanese population by analysing big data collected for patients.
Given Japan’s mature and considerable ICT market (worth nearly €370billion
45
), its
substantial ageing population, its high concentration of people clustered in urban areas,
and its tech-friendly society, the country currently gathers the set of conditions for
success and growth in developing telemedicine in the country. Indeed, an
increased focus on home care (including mobile health and telehealth) would alleviate the
country’s reliance on hospitals. Patients could receive proper care from home, and
hospital bed utilisation would be optimised for emergencies.
A high-quality technological network exists and can deliver telemedicine in Japan, with 3G
and 4G systems and high-speed broadband Internet widely available. The widespread
prevalence of Internet connectivity may have some impact on mobile health and
telehealth deployment in Japan. Mobile telemedicine solutions could be used for collecting
and measuring vital health information more consistently.
According to a 2014 survey by the Japanese Ministry of Health, Labour and Welfare
(MHLW), only 18 hospitals and 544 clinics nationwide offered telemedicine and telecare.
However, with technological advances, experts in deregulation urged the Ministry to
amend the legislation in force. The Ministry had prohibited telemedicine for a long time,
only authorising face-to-face treatment and making telemedicine available only to
patients with chronic diseases in remote areas (Medical Practitioners Act). A decision
issued in 2015, strongly backed by medical associations, and effectively lifted the ban,
allowing various start-ups to offer the service. This prompted many medical start-ups
to launch telemedicine services in metropolitan areas.
Finally, recommendations have been issued for implementation
46
regarding the revision of
the article 20 of the Medical Law, in particular on the clarification of who can practise
telemedicine, the enlargement of the scope of practitioners beyond medical doctors, and
the definition of reimbursement schemes and number of telemedicine services eligible for
reimbursement.
It is important to notice that most telemedicine studies in Japan are published in
Japanese, and thus, they are practically inaccessible to the rest of the world.
45
https://www.statista.com/statistics/820926/ict-industry-total-sales/
46
The American Chamber of Commerce in Japan (ACCJ) (2014).

Market study on telemedicine
Final Report
61
2.2. Market environment
2.2.1 Market Players
The literature suggests five main categories of telemedicine solutions providers:
telecommunications companies and mobile operators; big ICT and electronics groups;
manufacturers of medical/monitoring devices/platforms; pharmaceutical industries; and
start-ups. The Figure below shows these categories, along with example companies
47
,
48
,
49
. A more detailed presentation of each category follows.
Figure 31: Telemedicine value chain key players
Telecommunications companies and mobile operators
They actively invest in telemedicine to make up for declining market shares and
decreasing profit in their traditional activities. Medical products and services are seen as a
premium segment in which security and quality requirements outweigh price
considerations.
In addition to providing data centres to store and manage health data, telco players make
available networks with sufficient bandwidth and data upload capabilities, supporting
high-quality image and video-based services. This is crucial for services such as
teleconsultation and teleradiology, which demand high-resolution pictures. Therefore,
47
Androuchko L. Wright D., “Telemedicine and developing countries”, Journal of Telemedicine and Telecare, vol.
2, nº 2, 1996, RSM Press Ltd.)
48
Baum P., Abadie F., “Market Developments – Remote Patient Monitoring and Treatment, Telecare,
Fitness/Wellness and mHealth”, JRC Scientific and Policy Reports, 2013.
49
Commission Staff working document SWD(2012) 414 final on the applicability of the existing EU legal
framework to telemedicine services.

Market study on telemedicine
Final Report
62
telemedicine may increase traffic on their networks, thus boosting their revenues.
Also, remote patient monitoring services – despite requiring lower bandwidth – ensure
continuous cash flows.
Big ICT and electronics groups
Telemedicine market players like these rely on their core competences, such as managing
data and structuring workflows. Their strategy is to capture sizeable shares in this fast-
growing market.
More specifically, ICT market players use their expertise to create innovative products,
software and platforms that provide patients and healthcare professionals with
increasingly advanced functionalities. For instance, personal health record apps or
platforms on which people can access their medical records, track their data from devices
(smart activity, blood pressure monitors, blood glucose monitors) and share it with
anyone they choose.
Manufacturers of medical/monitoring devices/platforms
They now have a wider market scope, since they target the remote patient monitoring
market, home-based care, independent living and well-being. Consequently, they have
adapted their value proposition so that it meets non-professionals’ needs. Their products
usually incorporate advanced technology and include sensors, software and/or
connectivity to EHR so that consumers can consult the data collected.
The market seems highly fragmented, with numerous competitors (including sport
equipment manufacturers entering the market) getting a small share of it
50
. Brand loyalty
and improved technology are the main factors behind growing sales, even though the
market is already well established.
Pharmaceutical industries
Pharmaceutical companies see telemedicine as a great opportunity for growing sales,
especially in a context of toughening regulation and high R&D costs for developing new
drugs. Furthermore, generic producers are an additional threat to pharmaceutical
companies, as the latter need to justify the higher prices of their new drugs compared to
generic medication.
50
Some of the products available are ePatch (BioTelemetry), Health@Home, DiabMemory, myAirCoach,
Commander FLEX (Medtronic), Heartline ECG Monitoring (Aerotel), LifeWatch and Latitude NXT (Boston
Scientific Group).

Market study on telemedicine
Final Report
63
By deploying telemedicine, they are able to build new revenue streams. Partnerships
between pharmaceutical companies and ICT groups are multiplying. Their complementary
expertise enables them to offer cutting-edge products and services.
51
Start-ups
Telemedicine start-ups are proliferating. They provide tailored solutions and usually
aim at making easy access to healthcare the status quo. Their innovative solutions have
enabled them to easily raise funds; providing them with the financial means to deploy
their products. Innovative delivery models are a distinct part of the start-up service
officering, e.g. diagnosis through gaming or through the use of virtual reality.
52
Other types of telemedicine market players include universities, research centres,
and EU-funded projects, which also develop, test and deploy telemedicine solutions.
In recent years, the development of synergies between healthcare and technology has
generated a telemedicine market environment, both globally and in the EU, defined by a
set of key players, including the producers of products and services, their users, and the
regulating authorities. Figure 32 below provides a few examples of synergies between the
healthcare and technology industries, as illustrated by recently developed key
partnerships.
This intersection between healthcare and technology has given rise to numerous business
opportunities and benefits from the fast-paced diffusion of digital communication
technologies. As healthcare providers have begun to use technology for patient
management and care purposes, individuals around the globe have also started to adopt
wearables, biosensors and digital applications for health management and care. The
demand for technology-based treatment and care has allowed market players to deliver
telemedicine solutions at scale and with constantly increasing cost-effectiveness.
51
The collaboration between Roche and Qualcomm Inc., which gave birth to Qualcomm Life, is one of the many
illustrations of this. Qualcomm Life has developed the 2net Hub, a wearable medical device that transmits vital
medical data to the 2net platform for telemonitoring purposes. Servier has followed to same approach to
develop WeHealth.
52
Two examples to illustrate this point are Diapason and KineQuantum. Diapason (Immersive Therapy) is a
mobile application that performs accurate audiograms through games to locate the person’s hearing loss, then
offers therapy based on sending sound signals to readjust parts of the hearing. KineQuantum’s virtual-reality
headphones project users in 3D games, measure their movements rigorously and show their progress, as part
of physiotherapy.

Market study on telemedicine
Final Report
64
Figure 32: Examples of recent partnerships between traditional healthcare and technology
companies
Source: XERFI (2017)
At the same time, large ICT and electronics groups also invest in attractive start-ups
53
to gain a better market position and keep the pace of innovation in telemedicine.
2.2.1.1. Key players and investments
A significant share of healthcare technology and innovation in connected health is
increasingly driven by communication technologies focused on transmitting large data
streams (precision imagery, video, etc.). The market environment depends on the
dynamics of the digital health industry: market players in the areas of health analytics,
telemedicine, connected health devices, etc., have developed and established within this
market environment.
Numerous established firms, such as Medtronic, Roche, Johnson & Johnson, have been
actively pursuing investments during recent years, constantly reshaping the telemedicine
field.
54
53
For instance, IBM has recently acquired Cleveland-based Explorys, a healthcare intelligence cloud company
that has built one of the largest clinical data sets in the world.
54
XERFI. (2017). The Global Medical Technology Industry.

Market study on telemedicine
Final Report
65
This “expansion-oriented” behaviour amongst main market players can be explained by
the increasing investment trend in the field during recent years. The Figure below
indicates the amount of investments in digital health worldwide from 2010 to 2017
55
suggesting a sharp increase from 2014 to date.
Figure 33: Total digital health industry funding worldwide (2010 – 2017)
2.2.1.2. Consumers
Population ageing is accelerating worldwide, particularly in developed economies, where
fertility rates are down and life expectancy is rising. In 2015, people aged over 65 made
up 17% of the developed markets’ population. The increase in the average age of a
population results in a higher incidence of chronic diseases, thus creating sustainable
demand for health technologies
56
. According to the base scenario of the population
projections by Eurostat for the period 2015 to 2050: the share of people aged 65 and
over is projected to increase from 18.9% in 2015 to 28.1% by 2050, with the share of
people aged 85 and over more than doubling from 2.5% in 2015 to 6.0% by 2050
57
. This
poses specific challenges to the provision of medical services in a traditional set-up, also
for healthcare providers.
A second important trend is the worldwide increase in chronical diseases. As discussed
above, cardiovascular diseases, diabetes and chronic obstructive pulmonary disease are
the most treated by companies. Healthcare systems intend to limit the burden linked to
these conditions by prevention programs and better monitoring and long-term treatment.
2.2.1.3. Regulators
Governments in many countries worldwide are coming together to establish frameworks
that promote the convergence of standards and regulations for telemedicine solutions. For
55
Source: https://www.statista.com/statistics/388858/investor-funding-in-digital-health-industry/
56
XERFI. (2017). The Global Medical Technology Industry.
57
http://ec.europa.eu/eurostat/statistics-explained/index.php/Statistics_on_regional_population_projections
1,0
1,5
1,8
2,2
5,4
5,6
7,4
9,8
0,0
1,0
2,0
3,0
4,0
5,0
6,0
7,0
8,0
9,0
10,0
2010 2011 2012 2013 2014 2015 2016 2017
Percent

Market study on telemedicine
Final Report
66
instance, the Global Harmonization Task Force (GHTF) was initiated by a main group of
countries (the US, Canada, Japan, the EU and Australia), with the objective of
streamlining and harmonising all regulatory requirements regarding medical technologies.
In addition, efforts have been made at global level to help developing economies such as
India, China and Brazil converge in terms of regulation in the sector. These efforts led to
the creation of the International Medical Device Regulators Forum (IMDRF) in 2011, a
coalition of medical device bodies from various countries that seeks to accelerate global
regulatory convergence across the medical technology industry.
58
2.2.2. Supply and demand structure
Different forms of provision and use of telemedicine solutions and services can define the
supply and demand structure in the telemedicine market. On the supply side, the current
complexity and cost of providing telemedicine solutions and services is driving two
business models: a managed service model operated by service providers, and a
technology platform model operated by medical personnel. On the demand side, the
current use of telemedicine solutions and services is driven by the needs of institutional
and individual users to access different degrees of health services for expertise,
consultation and monitoring.
Three of the many drivers influencing IT investment in healthcare are ubiquitous access
to mobile technology, the criticality of IT security, and the rising need for chronic care
following the demographic and epidemiological transformation
59
. Today, healthcare
systems usually focus on hospital-centric care models and are often not well equipped to
meet today’s epidemiological challenges of preventing chronic diseases, diagnosing them
early and managing them effectively. Healthcare stakeholders across the EU are urged to
leverage innovative technologies to fundamentally redesign the way in which healthcare
is administered and delivered.
Technology is developed rapidly, although the adoption is slow mainly due to barriers to
the adjustment of societal behaviours. Society, especially the patient population, needs
time to adapt to the technology being offered. There are big gaps between the two
dynamics; this is mainly due to the lack of evidence on the efficiency and utility of
telemedicine. It is difficult to adopt if the different parties involved do not understand
each other. Indeed, the medical and paramedical professions do not often understand the
IT development professions, or the business models. This issue of integration of different
aspects of telemedicine makes it hard to for the offer to meet the demand. The complex
use of solutions requires a professional to communicate and facilitate the discussion
between technical people and health professionals.
2.2.2.1 Characteristics of the supply side
In recent years, the main players in the digital and health industries have greatly
increased their focus on healthcare technologies designed to provide remote access to
health services and improve patient management and monitoring. Nowadays, digital and
58
XERFI. (2017). The Global Medical Technology Industry.
59
Giguashvili, N., Alexa, J. IDC Health Insights (2016). CEMA Healthcare Outlook 2016: Transformation Under
Way.

Market study on telemedicine
Final Report
67
health industry players mainly provide telemedicine solutions and services through two
principal business models:
1. Managed services, through which a remote healthcare activity is outsourced by
an institutional user to a service provider. In this model, the service provider,
which is specialised in the technology embedded in telemedicine solutions, enables
functional telehealth care provision at a lower cost, based on the optimal
transmission and exchange of clinical data and the evaluation of results.
2. Technology platform services, through which a manufacturer or provider puts
in place the infrastructure to support the remote delivery of medical services.
Large medical institutions that offer remote medical services to individual patients
and other smaller institutions operate this infrastructure.
These two main business models encompass all different types of solutions and technical
types of telemedicine products and services described in Chapter 1. Indeed, either
through outsourcing or internalisation, healthcare providers rely on digital industry
players to set up and manage/support products, platforms or databases to ensure the
provision of remote health services.
Nevertheless, the current dynamics of the telemedicine market indicates that variations in
the provision of telemedicine solutions and services will converge into medical services
operated from technology platforms, where a group of technology providers will
manage/support remote care medical services. This convergence towards outsourced
telemedicine services from healthcare providers to digital industry players is explained by
the degree of specialisation required from the service provider, and the transaction costs
associated with either business model or the other.
Based on the interviews with key stakeholders in the value chain of telemedicine, it can
be concluded that the most predominant types of telemedicine solutions and services are:
Teleconsultation: in this case the doctor communicates remotely with the patient,
using for example video conference (with dedicated software that ensures privacy), to
hear the symptoms and make the diagnosis. Teleconsultation could also work well in
cases of regular prescription of drugs or medical tests (e.g. blood test) or just medical
advice on specific issues. However, lack of direct human interaction can be problematic
for some examinations or treatments (e.g. if the doctor need to inspect the ear). In
these cases, a face-to-face meeting with the doctor is a necessity. There are other
occasions where an initial physical meeting is important followed by teleconsultations
for monitoring the development of the illness. In essence, for diagnostic/consultative
context telemedicine can be widely adopted. However, treatment may still require a
physical visit the doctor in many occasions.
Telemonitoring: this type refers to digital therapeutics that can be used anywhere
(with the appropriate device and application). For example, a patient in a comma can
live at home and constantly monitored remotely by a hospital clinic. Telemonitoring
can also be important to lonely people with dementia or cognitive decline. Mobile
devises, wearables, smart homes, connected vehicles and advanced telemonitoring
devices including life support devices combined with technology such as the Internet of
Things, AI and Data analytics can enable remote healthcare and early preventative
intervention (already very advanced in the US) at a large scale.

Market study on telemedicine
Final Report
68
The development of technology provides numerous telemonitoring options. Biomarkers or
sensors of activity enable doctors to do a constant monitoring of the patient, look for risk
factors, and identify health conditions earlier. Sensors are providing adequate data and
input for clinicians to carry out assessments; these can provide early warnings about any
factor that goes wrong.
Currently, there are many options to monitor physical activity when doing sports or at a
patient’s home. Sensors mount on the walls can track in-house movement, which then
can be analysed in order to provide behavioural patterns that can be monitored. Such a
telemedicine application is useful for elderly people whose health may radically
deteriorate if they fall and get injured.
Today, solutions can provide telemonitoring for weak heart conditions of a patient on a
constant basis. As soon as there is an alert, the clinician is notified and can take a
decision that will help prevent the person from going into a cardiac arrest. These types of
solutions are currently being used in the US in order to reduce the risk of stroke with an
early preventive intervention.
It is also apparent from the interviews that while market players are growing very fast,
even though there is an unmet demand, the level of adoption is rather low. Thus, it is
important for the industry to better understand the client needs and meet the actual
demand from clinics and hospitals who ask for specific solutions. For a higher adoption of
telemedicine solutions, the clients (individuals, clinics, hospitals) need to be convinced
that that the solutions are suitable for them. If the clients have to pay for a solution,
which is not going to be reimbursed by the health care system, the decision to buy the
solution becomes harder. Thus, either the market players need to provide good value for
money solutions that will be easier to sell, or the national governments and the EU has to
cover some all the expenses to assist the further deployment of telemedicine.
One key barrier identified during the interviews is the apparent conservatism in the
adoption of new technologies in established practices. For example, many clinicians in
many countries are very conservative about adopting new tools and methods to do
therapy, even though they are aware of the benefits of telemedicine. This is a big barrier
in the clinical healthcare provider area. This may be due to lack of awareness of lack of
trust at the abilities of telemedicine to replace traditional approaches. Thus, interventions
are necessary to both raise awareness among health professionals and managers about
the benefits of the adoption of telemedicine the potential return of investment as well as
to ensure that telemedicine products and solutions available in the market are credible.
Furthermore, there are apparent difference among EU member states in terms of medical
care attitudes that also affect (promote or restrict) the digital transformation of health
care. The business model behind each system is different. In Sweden, the consumer
makes the choices, thus it seems to work better than other countries. In the Netherlands,
arguing to "keep patients away from the hospital" is counterintuitive as that means killing
the business. The more patients, the more procedures, the more money. The same could
be argued for other EU medical systems, where income is generated by having patients in
the hospitals and there is uncertainty for the sustainability of the turnover when a
significant part of treatment happens remotely. This is a key barrier also related to the
reimbursement model for telemedicine versus traditional medicine.
Changing the model is a long process. For example, it took 5 years for the DK authorities
to be convinced to change the model. The regulator must understand how the financials
work in a new model in order to be able to support the uptake of telemedicine.

Market study on telemedicine
Final Report
69
Another interesting area of this market relates to services that can offered to a big scale.
In this submarket, the biggest players are in diabetes prevention in the US. Omada
60
, “a
digital behaviour change program that can help the patient lose weight, reduce his/her
risk for chronic disease, and feel better than he/she have in years” is the biggest one.
Omada transformed diabetes prevention programmes to a digital therapy solution to
prevent diabetes. Using behaviour science the can help people change their living habits
(eating, exercising, sleeping), improve their health and reduce their risk of chronic
disease. They have been working to get a reimbursement status in the US and according
to their website this is the case for some health plans.
It is also apparent from the interviews that if the therapy from a telemedicine solution is
well documented, recognized and well established, it is possible for the State or the
health insurance to pay for it, but for that to happen it needs to have a strong clinical
evidence that the therapy actually works. Indeed, it is critical for any provider of digital
therapy to have clinical evidence that the solution actually works in order to get a
reimbursement model. In the meantime, what these market players can do is to get
clinics and hospitals to finance the solution from their internal budget. Many market
players and institutions who are willing to pay for it when they are convinced that the
solution actually works also apply this business model. Since healthcare systems are
often both care givers and service providers (take care of patients and get paid by
patients), they have a strong incentive to implement good solutions.
The issues around telemedicine based on the interviews conducted match the description
of services and solutions identified during the mapping exercise.
2.3.2.2. Characteristics of the demand side
The demand for telemedicine solutions in the market is mainly determined by two types
of users: individuals and healthcare providers. National States and their health care
systems are currently facing challenges due to demographic and epidemiological trends
that add pressure to meet the needs of ageing populations with an increasing incidence
and prevalence of chronic diseases. Regarding this particular aspect, the demand is – and
will continue to be – affected by the dynamics of chronic disease amongst the EU
population. According to the World Health Organization (WHO), over 85% of deaths in the
EU are due to five major chronic diseases (diabetes, cardiovascular diseases, cancer,
chronic respiratory diseases, and mental disorders).
61
Therefore, the potential demand for telemedicine applications can be analysed under a
market study approach that allows us to estimate the needs of individual and healthcare
providers under the aforementioned ageing and disease constraints. This study approach
must also integrate the potential willingness to pay for telemedicine solutions across EU
Member States and EEA countries, based on historical sectorial health data. This approach
relies on information available from Eurostat, most of which is included in the set of
European Core Health Indicators (ECHI) defined by the EU-funded Joint Action (JA) on
European Community Health Indicator Monitoring (ECHIM) in support of the EU Health
Strategy
62
.
60
https://www.omadahealth.com
61
Giguashvili, N., Alexa, J. IDC Health Insights (2016). CEMA Healthcare Outlook 2016: Transformation Under
Way.
62
See https://ec.europa.eu/health/indicators/echi/list_en

Market study on telemedicine
Final Report
70
The set of indicators used for the study of the potential demand is structured under the
three main pillars of the ECHI indicators: i) demography, ii) health status, and iii) health
interventions/services.
Demand estimate approach
- Step 1: The first step in our approach to study the potential demand is to
link information on the European population (total demography) with
information on the population presenting health conditions that require the
provision of healthcare services. Using information available from Eurostat
on the self-reported prevalence of specific diseases in the population across
countries, we estimate the number of individuals suffering from critical
health conditions to define the population at risk as a base for our demand
analysis. The outcome of this first step is an estimate of the potential
market size, expressed by the number of individuals affected by the
diseases that require the provision of health services.
- Step 2: The second step of the approach involves reconciling the total
population of individuals at risk (step 1 analysis) with information on
Member States’ total health expenditure across different types of
healthcare providers. This stage allowed us to initially segment the
potential demand for telemedicine solutions by distinguishing between
health expenditure by hospitals, outpatient healthcare, residential long-
term healthcare and other preventive healthcare providers.
- During this step, we estimate the potential market value for telemedicine
solutions, taking into account the population at risk and the associated
share of health expenditure. The outcome of the analysis is an estimate of
the potential expenditure that can be associated with serving the total
population affected.
- Step 3: The last step in our approach uses the output from the previous
analysis, combined with the parameter on the willingness to see a doctor
over video, which was obtained in the American Well survey (2016). This
provides an indication of a patient’s desire to be treated with the help of
telemedicine solutions, and is used as a proxy to indicate the extent to
which the total population is likely to shift towards telemedicine (along with
the associated expenditure), and hence reflects the potential future
expenditure in telemedicine solutions.

Market study on telemedicine
Final Report
71
Figure 34 : Online health utilisation – “In the last 12 months, how often have you used, if ever,
health and care services provided online without having to go to the hospital or doctor's surgery
(for example, by getting a prescription or a consultation online)?”
Source: Eurobarometer 460 “Attitudes towards the impact of digitisation and automation on daily
life”(2017)
63
It is important to note that the three-step approach can be expanded to take into account
specific chronic diseases by introducing an intermediary step between steps 1 and 2. In
this particular case, we would estimate the potential volume of patients affected by a
specific set of chronic diseases; the intermediary step of the approach would involve
estimating the proportion of inpatients treated by hospitals, and by chronic disease,
amongst all inpatients treated.
As part of this intermediary step, we would obtain a set of parameters, one for each
chronic disease, enabling us to estimate the share of healthcare expenditure that can be
associated with each of these chronic diseases. These shares could therefore be applied
as parameters under step 3 of the approach in order to obtain an estimate of the
potential market value for telemedicine solutions that takes into account the population of
individuals affected by the chronic diseases in question.
64
Demand estimate results
Under the first step of the demand estimation, we used the share of individuals suffering
from a long-standing illness or health problem drawn from the SIMPHS 2 survey. This
63
European Commission (2017).
http://ec.europa.eu/commfrontoffice/publicopinion/index.cfm/ResultDoc/download/DocumentKy/78998
64
It is important to highlight that in the absence of such information for all types of healthcare providers, this
analysis relies on a hypothesis of the proportions being distributed equally across providers and uses the
estimates drawn from hospital inpatients.
5%
6%
7%
81%
1%
18%
0%
10%
20%
30%
40%
50%
60%
70%
80%
90%
Once Twice Three times
or more
Never Don't Know Total 'At
least once'

Market study on telemedicine
Final Report
72
share was calculated using the survey data by country for 13 EU countries
65
, the
remaining countries in were assigned the average share.
The share of individuals suffering from a long-standing illness was applied to the total
population by country in order to obtain the volume of inhabitants possibly suffering from
a chronic disease. Results from this estimation indicates that in average, nearly 40% of
the EU population is suffering from a chronic disease. The Figure below provides an
illustration of the potential market distribution, expressed in volume (total inhabitants
suffering from a chronic condition), which could be addressed by the wide deployment of
telemedicine solutions.
Figure 35 : Estimated total population suffering from a chronic disease (in volume)
Source: PwC Analysis (based on Eurostat data on total population and SIMPHS 2 survey data on long-standing
illness)
Under the second step of the analysis, this distribution of the estimated population
suffering from a chronic disease (in volume) was multiplied by the estimated health
expenditure on chronic diseases. For this purpose, we used Eurostat information on
hospital discharges (records of at least one hospitalization day) by type of health issue,
focusing on any chronic disease. In average, hospital discharges associated to the
treatment of chronic diseases across the EU represent 11.6% of all hospital discharges.
This share was then applied to the total amount of health expenditure expressed in euros
per capita for each of the following types of healthcare providers:
Hospitals;
Residential long-term care facilities;
Providers of ambulatory healthcare;
Providers of preventive care.
65
Member States participating in the SIMPHS 2 survey: Austria, Belgium, Germany, Denmark, Estonia, Finland,
France, Italy, Netherlands, Sweden, Slovakia, Slovenia, Spain.
0
5
10
15
20
25
30
35
40
Million Inhabitants

Market study on telemedicine
Final Report
73
Multiplying the estimated number of individuals addressed by the market with the
estimated amount of health expenditure per individual associated with the treatment of a
chronic disease provided the potential market value addressed by the EU wide
deployment of telemedicine solutions. The Figure below illustrates the distribution of the
potential market value by country.
Figure 36 : Estimated health expenditure associated with the treatment of a chronic disease (in
value)
Source: PwC Analysis (based on Eurostat data on health expenditure and SIMPHS 2 survey data on
long-standing illness)
Under the final step of the estimation, we applied to each estimated market volume and
value the likelihood of consulting a doctor or a nurse online drawn from the SIMPHS 2
survey; the total share of individuals who declared that they are likely or very likely to
consult online amounts to 17,9% of the surveyed sample.
This result indicates that the potential market volume for the EU concerns about 36.6
million people and amount to over 10 billion euros for the sole EU territory. The Figures
overleaf illustrate the distribution of potential market volume and value for the EU under
current condition, estimated using the likelihood of consulting a doctor or a nurse online
across the EU.
0
2000
4000
6000
8000
10000
12000
14000
16000
18000
20000
Million Euros

Market study on telemedicine
Final Report
74
Figure 37 : Estimated market demand for telemedicine solutions (in volume)
Source: PwC Analysis (based on Eurostat data on total population and SIMPHS 2 survey data on
long-standing illness and likelihood of consulting online)
Figure 38 : Estimated market demand for telemedicine solutions (in value)
Source: PwC Analysis (based on Eurostat data on health expenditure and SIMPHS 2 survey data on
long-standing illness and likelihood of consulting online)
0
1
2
3
4
5
6
7
Million Inhabitants
0
500
1000
1500
2000
2500
3000
3500
Million Euros

Market study on telemedicine
Final Report
75
2.2.3. Industry structure and market conditions
2.2.3.1 Availability and access to telemedicine solutions
Telemedicine solutions have recently started to take up across EU Member States. The
pace at which they are deployed depends on several factors, including the availability of
funding for investment in medical technology and infrastructure, the availability of digital
skills amongst medical personnel, and the ability of the market environment to match the
supply and demand for telemedicine solutions and services.
The rising costs of disease treatment within national health systems and the
demographics in the EU are increasingly putting pressure on public expenditure budgets;
consequently, cost containment and efficiency is becoming a great concern, both for EU
Member States and worldwide.
Relevant authorities are now closely scrutinising healthcare spending, with the objective
of reducing and optimising expenditure in the provision of health services. With constantly
increasing demand for health services driven by demographics and epidemiology, and
lower relative funding for health systems, diffusion and access to telemedicine services
will increasingly become a major strategic concern in the short term.
To address this issue, key players in the technology and medical sectors are designing
and providing holistic solutions, comprising both devices and solutions (e.g. software and
consulting), to improve patient outcomes while maximising care-setting efficiency.
66
The
wide-scale availability of these healthcare services will mainly depend on the ability of EU
and national authorities to adapt and adjust to convenient models that match the supply
and demand of telemedicine solutions. For instance, of the way in which telemedicine
costs are reimbursed needs to be thought out. Either fee-based or value-based
reimbursements will need to be studied, taking into account the different ways of
providing and consuming telemedicine solutions, whilst considering the objective of
improving the quality of health services at a lower relative cost for society.
Finally, a lack of willingness to adopt new solutions is a barrier to innovation. Resistance
to change is a complex issue related to various factors, including the problems of an
ageing workforce, salary levels, workloads, and often the lack of digital skills. In
particular, the shortage of necessary digital skills among clinical personnel is viewed as
one of the main factors hindering the uptake of telemedicine solutions and services across
the EU, as well as being a key barrier to the uptake of e-health innovation.
2.2.3.2 Financial sustainability of health systems across Member States
Several factors need to be considered regarding the reimbursement models associated
with the future deployment of telemedicine solutions and services across the EU, most
notably the escalating costs associated with healthcare provision (infrastructure and
operational expenditure) and the dynamics of the workforce in the medical field.
The availability of financial resources for healthcare is extremely problematic in some
CEMA countries. According to the latest available data from the World Bank, the average
amount spent on healthcare in 2013 was €689 in Central Europe and the Baltic States
and €2,595 in the EU overall. However, healthcare systems continue to struggle with
66
XERFI. (2017). The Global Medical Technology Industry.

Market study on telemedicine
Final Report
76
unsustainable conditions due to demographic dynamics in the EU, the decreasing share of
the active population, and the increasing need for treatment for chronic diseases.
These conditions continue to put pressure on healthcare costs. For instance, long-term
projections show that the fiscal impact will be high in most EU Member States. According
to the 2015 Ageing Report,
67
economic and budgetary projections for the 28 EU Member
States (2013-2060) indicate that the projected change in strictly age-related public
expenditure will amount to 2 percentage points of the GDP between 2013 and 2060, with
an increase driven mostly by healthcare and long-term care spending.
Healthcare financing systems will thus be challenged by the fact that complementing
healthcare funds with private expenditure (e.g. out-of-pocket payments and patient co-
payments for doctors’ visits) has proven to be extremely difficult, as these systems have
traditionally been funded primarily by public sources.
An additional factor to consider is that the human resources deficit in the medical sector
remains critical in certain Member States and healthcare systems, hindering healthcare
reach, quality and outcomes. The deficit of medical professionals is severe: on average,
there are currently only 3.5 physicians available per 1,000 inhabitants in the EU, despite
considerable government efforts and investment in training and educating healthcare
professionals.
This lack of healthcare workers needs to be considered with particular attention in central
and eastern European countries, which have a lower number of physicians per capita than
those in Western Europe. For example, in 2015 Romania has 2.77 physicians per 1,000
inhabitants, and Poland has only 2.33, compared to 4.14 in Germany and 5.1 in
Austria
68
. Beyond the issue of human-capital availability, other considerations need to be
taken into account. The mix of an ageing population and an ageing healthcare workforce
indicates that while demand for medical services will grow, the supply of available skilled
labour will decline.
The factors affecting the production and uptake of telemedicine solutions must be
investigated to develop optimal pricing strategies across the EU and to allow supply and
demand in the industry to be matched.
2.3. SWOT analysis of the market
An increasing number of healthcare providers are starting to adopt connected
telemedicine technologies, as the sector has the potential to deliver significant cost
savings for healthcare provision across EU national health systems. Large medical
technology players such as Medtronic and GE Healthcare are seeking to build up
capabilities in the area by acquiring digital start-ups or teaming up with technology giants
such as IBM or Philips, which are currently adapting fast to tap the potential benefits of
digital health growth.
69
67
European Commission (2015). The 2015 ageing report.
68
http://ec.europa.eu/eurostat/tgm/table.do?tab=table&init=1&language=en&pcode=tps00044&plugin=1
69
XERFI. (2017). The Global Medical Technology Industry. The Market, (April, 2017).

Market study on telemedicine
Final Report
77
Figure 39: Potential economic impact of connected healthcare in the US
Source: XERFI, 2017
Business development in the sector rests on several factors, which we have considered
below as part of a SWOT analysis:
Strengths
Heavy political focus on healthcare as a core state responsibility.
Government support for global convergence of standards and regulations on medical
technologies, hence telemedicine.
Continued economic development in emerging markets outside the EU and converging
economies within the EU.
Intellectual property protection.
Weaknesses
Amongst the weaknesses of the EU in terms of telemedicine adoption, one may highlight
the need for more support and quantity of applied research projects to provide evidence
and properly reflect the actual benefits of the telemedicine. There is a need to increase
the base of evidence through implementation exercises as today we focus mostly on
theoretical studies, but lack actual large-scale implementation. Indeed, there is a
mismatch driven by the development of innovations that do not take into account the
actual needs of the patients. This mismatch reflects the different perspective from
different parties involved in the process of telemedicine adoption highlighting problems of
communication, collaboration, and lack of understanding between them. For instance,
nurses and doctors may have a different understanding of a patient’s need; as an
example, in the case of Parkinson’s disease, nurses are prone to focus on the quality of
life, while doctors are prone to focus on medication to be able to walk. In addition,
technicians and developers of telemedicine solutions will focus on the technical
development rather than the integration of technology across several professions. This
issue points out to the fact that today, telemedicine development and uptake is dealt with
from different angles depending on each party. When the come together they have a
better understanding of each other. To solve this weaknesses of integration all actors

Market study on telemedicine
Final Report
78
involved need to come together to clearly understand the medical professions, the
patient’s needs, and the technical development limitations.
Indebted healthcare systems increase pressure for cutbacks in healthcare spending.
Reduction in individual disposable income.
Increasingly stringent regulations, which can slow down technology diffusion and
adoption by wide pools of users.
Global inconsistencies in regulations.
Stricter and lengthier regulations increase time-to-market and development costs.
Lack of interoperability due to fragmentation.
Opportunities
Ageing and wealthier populations.
Limited access to healthcare in rural areas
Rising urbanisation is accompanied by a growing prevalence of lifestyle diseases that.
Opportunities in healthcare technology spurred by new digital technologies (5G, big
data and artificial intelligence, cybersecurity).
The market has high potential for growth with a significant likelihood of start-ups and
large firms entering the telemedicine market.
Threats
There are several threats or delaying factors in the EU concerning the uptake of
telemedicine. One of the key threats is the Global Data Protection Regulation, which is
generally positive, as it has streamlined the rules on the use of individual data, but has a
clear downside in terms of delaying the creation of evidence and the adoption of digital
health solutions. Indeed, the burden on small telemedicine start-ups is pushing these
companies leave the EU and settle in the US since the regulation there is more flexible
and allows them to work directly with large health systems. The uptake of telemedicine
depends on the handling and treatment of medical data, which requires permissions and
flexibility to carry out advanced analysis in order to generate evidence for the market a
convenient pace.
Other important threats for the uptake of telemedicine in the EU concerns regulatory
approval (by CE Mark) which is necessary, but is unaffordable for small companies.
Indeed, today, approval for a class 2 or 3 device the process becomes so expensive that
the process crowds start-ups out, limiting innovation as only big players can go through
the process. The only way to succeed is to have very strong support from big partners
(VC capital). Those pathways can work efficiently, although they will definitely hinder
disruptive innovation.
In addition, further threats include:
Cybersecurity risks posed by mobile and digital-related health.
Shrinking working population.
There may be significant rivalries due to the many major players in the sector
concentrating the market; new entrants are competing to gain a market share.
Bargaining power:
70
70
Technavio report, 2015

Market study on telemedicine
Final Report
79
- Bargaining power of suppliers: Several suppliers are active in the
telemedicine market, but they are not price-makers. End-product suppliers
and institutional users can reduce the suppliers’ bargaining power.
- Bargaining power of buyers: There are several successful and
established market players in the market as well as institutional consumers.
This allows for significant consumer market power.

Market study on telemedicine
Final Report
80
2.4. Company profiles
This section is dedicated to the key players operating in the global telemedicine market
71
.
It shows the positioning of some of these actors in Europe, complements the analysis on
distribution of services, and gives insight into solution portfolios. It further presents a
business overview of each player.
General
overview
Royal Philips is a leading health technology company focused on
improving people's health and enabling better outcomes across the
health continuum from healthy living and prevention, to diagnosis,
treatment and home care. It is the leader in diagnostic imaging, image-
guided therapy, patient monitoring and health informatics.
Philips telemedicine solutions target both patients and doctors, and have
demonstrated the following results:
Reduction in overall costs of care
Reduction of hospitalisations
Reduction of the number of days in hospital
Reduction of readmission rates
Solution
portfolio
Remote patient monitoring solutions
Remote chronic disease management solutions
Readmission management solutions, via home devices
Sleep therapy solutions
Geographical
reach
Global
Business
strategy
Philips’s strategy focuses on a more connected, predictive and
personalised care delivery. It prioritises partnerships; at present, the
company has 40 long-term relationships with healthcare providers from
medical universities to hospitals.
Recent
developments
January 2018: Strategic partnership with American Well™, the leading
U.S. telehealth provider
Deployment of Philips Avent uGrow, a parenting app to monitor baby
development and 24/7 access to professional medical consultations
HealthSuite Digital Platform: consumers and patients can secure and
select which data to share with health professionals
Philips will join the American Well Exchange™ clinical services
marketplace, which enables healthcare partners to exchange
telehealth services with one another and redistribute them to new
patient populations
71
As referenced in the Tender Specifications for the study, and in the market study “Telemedicine Market -
Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2014 - 2020” by Transparency Market
Research.

Market study on telemedicine
Final Report
81
General
overview
Polycom (now a part of Plantronics) is a provider of communications and
collaboration technology. More than 400,000 companies and institutions
worldwide defy distance with video, voice and content solutions from
Polycom.
In healthcare, Polycom provides video collaboration solutions that
connect healthcare professionals with each other and/or with patients.
The company also provides medical education, healthcare administration.
Solution
portfolio
Video collaboration solutions
Medical education and healthcare administration videos
Geographical
reach
Global
Business
strategy
Polycom solutions are flexible, i.e. designed to be applicable in any
environment, feature multi-vendor interoperability and have a high
degree of security. Similar to other key players, the company relies on
strategic partnerships for its success, including with: Huawei, Alcatel,
Cisco, Avaya, and Microsoft.
Recent
developments
January 2018: Merger with Obihai Technology, Inc., an innovator in
VoIP audio solutions
Cloud services launched focusing on audio devices and solutions
today, anticipating support to video communication by end of 2018
Polycom has unveiled new, high-end video capabilities, such as facial
tracking, dual monitor support, superior audio coverage, and easier
content sharing
General
overview
OBS Medical is the global leader in the development and provision of
predictive algorithms for identifying critical instability in patients that
is not picked up by traditional methods. As such, the company’s main
customers are healthcare professionals.
Solution
portfolio
Medical software
Geographical
reach
Global

Market study on telemedicine
Final Report
82
Business
strategy
OBS Medical’s strategy relies on direct collaboration with healthcare
providers, hospital data service providers and medical device
manufacturers (e.g. ExcelMedical, Connexall, Caretaker Medical).
Innovation is an essential part of the company’s DNA – indeed, it
pioneers artificial intelligence to provide healthcare professionals with
patient risk stratification and alerts.
Recent
developments
OBS Medical aims at expanding in the US
General
overview
InTouch Health provides cloud-based network and virtual care solutions
that ensure connectivity for health systems, providers, and
patients at all times.
Over 130 health systems are supported by the company at present. It
boasts 8,600 registered network users and 1,000,000 telehealth virtual
care sessions over its platform.
Solution
portfolio
Virtual medical care platform
Telehealth devices
Geographical
reach
Global
Business
strategy
In addition to a portfolio of solutions, InTouch also offers support
services for telehealth implementation and consulting services. Its
goal is to expand its market share as a preferred partner for hospitals
and health schemes.
Strategic partnerships include those with: Bon Secours, hospitals,
Dignity Health, Ohio State University, Mission Health, the Hospital
Consortium of America, Standford Medicine, Kaiser Premanente, and
PinnacleHealth.
Recent
developments
April 2018: InTouch Health acquired REACH Health, a telemedicine
software company based in Georgia
January 2018: InTouch Health acquired Truclinic, a web design
telemedicine provider based in Utah specializing in direct to consumer
virtual care solutions

Market study on telemedicine
Final Report
83
General
overview
Honeywell Life Care Solutions (formerly Honeywell HomMed) offers
remote patient monitoring services, remote patient management
applications, as well as decision support and evidence-based disease
management.
Honeywell telemedicine solutions target healthcare providers, and have
demonstrated the following results:
Multimillion cost savings
Reduction of readmission rates
Solution
portfolio
Solutions integrating EHR and other medical records
Remote clinical monitoring software
Remote patient monitoring software and devices
Telehealth platforms
Geographical
reach
North America
Business
Strategy
Honeywell is one of the pioneers in telehealth for over 18 years. Its
success is based on quality but also lower product costs. Key
partnerships that have facilitated its market rise include those with:
MobileHelp, Samsung and Fuwe.
Recent
development
June 2017: Launch of latest version of the monitoring software,
including a more efficient navigation, a mobile platform, an improved
patient dashboard, advanced scheduling and reporting
Updates to the company’s telehealth platform featuring fully-
integrated video communication enabling individuals with chronic
conditions to meet with their doctors and nurses face to face without
having to leave their home
Honeywell Select Services introduced to streamline the telehealth
delivery process between healthcare providers and patients, by
monitoring patients from Honeywell’s nurse call center headquarters
General
overview
Cisco delivers ‘care at a distance’ solutions, connected imaging
solutions, telehealth and collaboration solutions. The company has
been in healthcare for more than 20 years, spanning 17,000 healthcare
organisations and 118 countries around the world.
In addition lower readmission rates, Cisco telemedicine solutions used by
healthcare providers have resulted in a lower numbers of adverse drug
events.

Market study on telemedicine
Final Report
84
Solution
portfolio
Virtual health solutions (telemonitoring, teleconsultation, video care)
Patient engagement solutions (e.g. Inpatient Bedside Technology)
Converged clinical workflow solutions (e.g. location tracking)
Health data exchange platforms and real time analytics
Geographical
reach
Global
Business
strategy
What differentiates Cisco is the company’s commitment to data
security. Indeed, Cisco offer secure access to network resources and
applications from any location in order to promote security best practices
that meet regulatory compliance goals.
Recent
developments
Goal to create a complete digital strategy for healthcare providers
around the world
Launch of HealthPresence Telemedicine Solution – an advanced video
collaboration technology giving patients an immersive and highly-
secure remote healthcare experience
Telemedicine pilot programmes in several countries to test willingness
to use and satisfaction with remote patient care
General
overview
Medtronic is a medical device company that acquired Cardiocom in 2015.
Cardiocom used to provide telemedicine solutions for daily remote
patient monitoring and disease management.
Used by healthcare professionals since 1998, Medtronic solutions have
recorded over 5 million telehealth patient months of use, and currently
service over 95,000 patients.
Solution
portfolio
Remote patient monitoring solutions, including advanced medical
monitors
Wireless cardiac monitors and mapping solutions
Remote monitoring system for diabetes (including insulin pump
systems, infusion sets, injection ports)
Geographical
reach
Global
Business
strategy
Medtronic targets specific diseases, in particular heart failure,
Parkinson’s disease, obesity, diabetes. In doing so, it establishes targeted
partnerships, for instance with IBM Watson.
Recent
developments
One of the company’s priorities is reaching underserved populations and
the Middle East where it actively works to expand clinic-based models

Market study on telemedicine
Final Report
85
General
overview
AMD Global Telemedicine is a pioneer in clinical telemedicine equipment
and technology that is used to connect a patient with a remote
healthcare provider.
AMD solutions primarily target rural health clinics, school health centers
and pharmacy clinics. To date, the company has over 8,300 patient end-
point installations set up in 98 countries.
Solution
portfolio
Telemedicine carts and systems
Telemedicine encounter management software (for real-time remote
patient exams)
Medical devices and equipment designed to deliver superior quality
medical images and precise patient data, especially for difficult or
remote application
Geographical
reach
Global
Business
strategy
AMD’s niche is rural and underdeveloped regions around the world.
Therefore, the company prioritises partnerships with local and national-
level public organisations, e.g. schools (health) alliances, national
telemedicine associations and national health associations.
Recent
developments
April 2018: Development of OnDemand Visit, a direct-to-consumer
telehealth platform
2017: 6
th
enhancement release of AGNES Interactive telemedicine
software that aggregates medical device data and shares it in real-time
with the remote physicians
General
overview
Allscripts provides hospitals and other healthcare providers with practice
management and EHR technology.
At present, it reaches 45,000 physician practices; 180,000 physicians;
19,000 post-acute agencies; 2,500 hospitals; 100,000 electronic
prescribing physicians; 40,000 in-home clinicians; and 7.2 million patients.
Solution
portfolio
Health management platforms
EHR platforms
Patient engagement platforms (notably FollowMyHealth)
Geographical
reach
North America
India
Australia

Market study on telemedicine
Final Report
86
UK
Singapore
Israel
Business
strategy
The strategy of Allscripts is based on two pillars: IT management
services as part of the value proposition and innovative technology.
Recent
developments
Goal to create the healthcare IT architecture of tomorrow and keep
being the industry leader enhancing excellence
Selected the Vidyo.io platform from Vidyo to power virtual consultations
directly embedded into the Allscripts FollowMyHealth patient portal,
thereby enabling patients to do video consultations with physicians
either over the web or through a mobile app
Series of acquisitions (e.g. patient communication and engagement
platform HealthGrid) in a shift to value-added care tools beyond its
current EHR-centric solutions
Collaboration with Hale Health on a free telemedicine solution for
sharing photos and videos, conducting live video visits and
asynchronous messaging
General
overview
GlobalMed is the worldwide leader in telemedicine enabling more than 3
million teleconsultations annually.
Solution
portfolio
Telemedicine stations
Examination cameras
Connected medical devices (conference cameras, stethoscopes, exam
cameras)
Video conferencing and software that allow for connected care and
dynamic collaboration of healthcare professionals at remote locations
Geographical
reach
Global
Business
strategy
GlobalMed relies on a number of key partnerships for its success,
including with leading medical groups, healthcare enterprises and
government agencies.
HP, TeleMedGlobal, AT&T, and BT are also among its business partners.
Recent
developments
GlobalMed recently acquired TreatMD, a telemedicine company that
provides “on-demand healthcare platforms”, via which patients can book
phone and video consultations with physicians.

Market study on telemedicine
Final Report
87
3. BARRIERS TO ACCESS TO TELEMEDICINE SOLUTIONS
Key takeaways
Telemedicine barriers exist in all countries but do not affect them to the same
degree. It is difficult to quantify how their impact varies from one country to the
next.
Since telemedicine is a multi-stakeholder market, barriers also affect the players
differently within the countries.
The lack of a legal framework means there are other underlying obstacles
(reimbursement, lack of interoperability, lack of acceptance).
Funding and financial incentives are key drivers of telemedicine initiatives.
From our mapping, the lack of acceptance of telemedicine solutions by stakeholders,
the poor regulatory framework, the insufficient funding and the inadequate IT
infrastructure are the most prevalent barriers to telemedicine widespread
deployment.
The aim of the chapter is to:
Identify and examine barriers to telemedicine in each country;
Highlight the main barriers encountered in the EU countries;
Analyse and interpret the areas where EU cooperation or action is needed, based on
existing literature;
Perform a SWOT analysis of the telemedicine framework; and
Provide policy recommendations for each barrier/dimension of telemedicine.
3.1. Identification and analysis of telemedicine framework conditions
The illustration below presents seven different types of conditions that create barriers to
telemedicine. A detailed presentation of each type follows.

Market study on telemedicine
Final Report
88
Figure 40: Different types of conditions creating barriers to telemedicine
Figure 41 below brings out the most important barriers hampering the use of eHealth
tools in programs within the framework of the ICARE4EU project. The study was
conducted in 2016 providing an online questionnaire to the managers of the 58 care
programs from 24 European countries. The results observed from the study are in line
with our findings in the mapping. All the barriers mentioned by the respondents were
confirmed by conclusions of publications and report (see Excel file that maps out the
barriers to telemedicine country by country).

Market study on telemedicine
Final Report
89
Figure 41: Barriers hampering the use of eHealth tools included in the programs (% agreeing)
Source: Melchiorre M.G., Papa R., Rijken M., van Ginneken E., Hujala A., Barbabella F., eHealth in
integrated care programs for people with multimorbidity in Europe: Insights from the ICARE4EU
project, 2016
3.1.1 Cultural conditions
Healthcare professionals consider telemedicine as a threat to the patient-doctor
relationship.
Stakeholders in more advanced countries (in terms of adopting telemedicine solutions)
need to communicate and raise awareness of the benefits of telemedicine. They are
crucial to demystify and popularise telemedicine practices in other countries.
72
A high level of mistrust towards technology in healthcare has been observed across EU
countries.
Lack of acceptance by doctors
A persistent cultural barrier impeding the wide adoption of telemedicine is the lack of
acceptance of using telemedicine practices by doctors and healthcare professionals in
general. A survey on the usability of telemedicine application among a few hundred adults
in Austria in 2015 identified the “lack of acceptance by doctors” as the second top ranked
overall barrier of the adoption of telemedicine.
73
It is somewhat related to the reluctance
to use innovative technologies to treat patients. The doctors’ attitude towards
adoption of telemedicine will strongly influence its acceptance by their patients. Thus, it
remains crucial to inform and train healthcare professionals about the advantages of
telemedicine to encourage wider deployment of telemedicine.
Besides, surveys conducted by polling organisations
74
highlighted that the doctor-
patient relationship was the prime emotional factor for healthcare staff. Regular
72
SIMPHS 2 (2013), JRC
73
Haluza D., Naszay M., Stockinger A., Jungwirth D. “Prevailing Opinions on Connected Health in Austria:
Results from an Online Survey”, Int J Environ Res Public Health, August 2016
74
Ipsos-MORI, “What Matters to Staff in the NHS”, 2008
60%
55%
55%
53%
52%
50%
45%
40%
35%
33%
26%
0% 10% 20% 30% 40% 50% 60% 70%
Inadequate funding
Compatibility between eHealth tools
Inadequate technical/ICT support
Inadequate ICT infrastructures
A lack of skills among patients
An inadequate legislative framework
A lack of skills among care providers
Uncertainty about cost efficiency
Privacy issues
Resistance by care providers
Cultural resistance

Market study on telemedicine
Final Report
90
physical contact with patient is what stimulates most doctors in the way they practice.
Consequently, they might perceive telemedicine as a threat to a preferred way of
delivering health.
75
In addition to the fear of patient loss, the resistance from medical personnel to adopting
technology in healthcare can also be explained by conservatism in some countries.
Healthcare professionals tend to protect the traditional models they have grown up with
instead of embracing new ways of providing healthcare.
Lack of acceptance by patients
Figure 42: Factors explaining the lack of acceptance of telemedicine solutions by patients
Inadequate technology
Patients are sometimes reluctant to use telemedicine because they consider the existing
technology inadequate for treatment; this is prevalent in countries with limited access
to/penetration of advanced technology. In these cases the reason for the patient’s
reluctance to adopt or even demand the use of telemedicine solutions is lack of
awareness of the existence of advanced solutions or low confidence in technology in
general.
Inadequate digital skills
The lack of acceptance by patients is also linked to the low level of digital literacy in
parts of the population in some countries. People who live well below the poverty line and
older people aged over 65 or over 80 often have low ICT skills and are unfamiliar
with/unaware of new technologies. In 2016, 17.3% of the population in the EU were at
risk of poverty and 7.5% of the population in the EU were severely materially deprived
76
.
In addition, 27 million people aged 80 and over lived in the EU in 2016. Finally, 169
million Europeans between 16 and 74 years, this is 44% of the population, do not have
75
SIMPHS 2 (2013), JRC
76
https://ec.europa.eu/eurostat/statistics-explained/index.php/People_at_risk_of_poverty_or_social_exclusion

Market study on telemedicine
Final Report
91
basic digital skills
77
. In all these cases, the lack of digital skills can be an important
reason of weak end-user adoption of telemedicine solutions.
Preference for personal contact
Some EU nationals strongly believe that technology will never act as a substitute for
physical contact in health. In some cases, a deep-rooted emotional bond links
patients and doctors. General practitioners or other healthcare professionals have
sometimes been family doctors for decades and patients want to keep this relationship
the way it has always been. In health, old habits die-hard.
Data security
Another reason for the lack of acceptance by patients is the general fear of data
security breaches. People are not eager to store and share their medical data online
because they fear that their personal information might be disclosed to third parties.
Because of hacking risks, patients are still reluctant to share their data in most countries.
Suspicion of corruption within the healthcare system
Finally, in a few countries it emerges that the suspicion of corruption within the
healthcare system is a major barrier to telemedicine deployment.
All these factors are impeding the complete development of telemedicine. This resistance
can even turn into fierce opposition. For instance, the European electronic health
insurance card
78
was one of the regulatory actions in Lead Market Initiative
79
but the
initiative was hindered due to national opposition from Member States.
80
The unshakeable doctor-patient relationship
Opponents to telemedicine often blame ICT for dehumanising healthcare and fostering
impersonal disease management. Technology should not interfere with the sacrosanct
doctor-patient relationship.
Patients and doctors are really attached to their usually longstanding personal
relationship as sometimes doctors provide also psychological support to their patients.
Telemedicine, with technology acting as an intermediate, is perceived to potentially
jeopardise that relationship. Primary care is firmly rooted in face-to-face
interaction.
Based on the findings from the Ipsos MORI study
81
, participants perceived that financial
interests rather than humane considerations drove the introduction of ICT in healthcare.
Yet, the latter constitutes the prime emotional motivator for healthcare professionals.
In summary, the feeling that telemedicine endangers the doctor-patient relationship is a
barrier to these services being mainstreamed. Face-to-face interaction is still a pillar of
healthcare today for both patients and healthcare professionals.
To overcome these cultural barriers, pressure from the demand side, communication and
training are potential actions to pursue.
77
https://ec.europa.eu/digital-single-market/en/news/digital-skills-gap-europe
78
http://ec.europa.eu/health/ph_information/implement/wp/systems/docs/ev_20071119_co01_en.pdf
79
http://ec.europa.eu/growth/content/lead-market-initiative-%E2%80%93-speed-time-market-innovations-
and-pilot-new-innovation-policy-0_en
80
Final Evaluation of the Lead Market Initiative
81
Ipsos MORI, Attitudes to healthcare services in the UK, 29 November 2013. Ipsos MORI interviewed a
representative sample of 1,009 adults aged 18+ across Great Britain. Interviews were conducted by telephone
between 12th – 14th October 2013

Market study on telemedicine
Final Report
92
3.1.2. Regulatory and policy conditions
Figure 43: Regulatory and policy barriers
The absence of a national strategy
The absence of a national-level strategy for telemedicine is unusual but observed in a
few countries. Some governments do not feel the urgency to develop telemedicine
and do not consider it a high priority of health strategic direction.
Lack of a legal framework
Most EU countries have a national strategy. However, it emerges that almost all lack a
precise legal framework to regulate telemedicine practices. Existing laws are
usually no longer in line with recent telemedicine innovations.
Thus, it is important for decision-makers to undertake reforms of the current laws in
order to promote telemedicine as this will also will encourage the local market. For
instance, in Switzerland, the government enacted a law in April 2017 to adopt
interoperable patients’ electronic health records (EHRs) and thus paved the way to its
broad adoption.
Interoperability – Lack of standards and guidelines
The lack of widely accepted/adopted standards and procedures represents a further
obstacle.
82
It limits trust in the quality and reliability of telemedicine solutions.
Within and across countries, telemedicine practices are not necessarily standardised and
thus not necessarily compatible. However, as mentioned above, there are initiatives in
these directions (e.g. the guidelines adopted by the eHealth Network), which should be
taken into consideration by the telemedicine market stakeholder even if these guidelines
are optional.
Many specific topics need special attention. For instance, no European rules address the
regime of medical liability or the standard of care for healthcare providers.
It brings about a serious issue of interoperability between telemedicine solutions.
Interoperability is fundamental to avoid legal obstacles (various telemedicine laws),
operational obstacles (various methods for data collection) or language obstacles (various
terminology or translation issues). The EU has taken measures to improve
82
Berti P., Verlicchi F., Fiorin F., Guaschino R. and Cangemi A., The use of telemedicine in Italian Blood Banks: a
nationwide survey, 2014

Market study on telemedicine
Final Report
93
interoperability and standardisation in eHealth
83
, but it still needs to develop a
uniform set of norms to regulate it. For now, Member States still have jurisdiction to
regulate this area.
84
Despite repeated initiatives from the EU to initiate coordination,
Member States have legal frameworks, approaches and levels of telemedicine
development that are too heterogeneous to hope for effective standardisation of practices
in the short term. Besides, countries sometimes adopt or adapt specific international
standards according to their own needs, which represents an additional barrier to
interoperability.
Through the eHealth Action Plan 2012-2020
85
, the European Commission aims to support
patients and healthcare workers, to connect devices and technologies, and to invest in
making medicine more personalised. In particular, by capitalising on tablet and
smartphone technology (mhealth) the Action Plan seeks to ensure the provision of
smarter, safer and patient-centred health services in the future.. In addition, digital
health is one sector of the Digital Single Market (DSM), which is one of the European
Commission's main priorities. In this direction, the European Commission adopted an
action plan in order to enable the digital transformation of health and care in the Digital
Single Market on April 2018. The aim of this action plant is to put EU citizens at the
centre of the healthcare system
86
. To allow EU wide deployment of developed solutions,
interoperability is high priority of the EU strategy in DSM. The eHealth European
Interoperability Framework references standards but Member States can choose to
approve open international standards. Therefore, non-interoperable solutions persist and
impede the scaling-up of telemedicine.
This lack of standards has mostly been felt in relation to data ownership and data
sharing. Indeed, countries have been struggling to implement regulations or
requirements related to cross-border sharing of patient data. Therefore, the need for EU-
wide harmonised standards and guidelines to ensure interoperability in data access and
processing has been explored in EU-funded projects (such as ESPOS and Antelope).
Data security
Because of the legal vacuum regarding data protection and security in most countries,
many fear a commercial or malicious use of patient data. The recent example of the
personal data misuse by Cambridge Analytica, which caught the public’s attention,
increased this fear. Determining the right of access to patient information is a difficult
question to solve.
87
How much patient information should be made available to hospitals?
Legal loopholes are persisting on these issues and responsibilities are not clearly
defined
88
. Sharp rise in hacker attacks and in medical identity theft has been noticed. Yet,
we underlined earlier that these concerns represent a major barrier to patients’
acceptance of telemedicine.
While policies are too permissive or non-existent in some countries, others have
adopted very stringent data protection laws, which impede any information sharing
between healthcare professionals. EU countries definitely need to strike a balance
83
https://ec.europa.eu/digital-single-market/en/interoperability-standardisation-connecting-ehealth-services
84
Vera Lúcia Raposo, Telemedicine: The legal framework (or the lack of it) in Europe, 2016
85
https://ec.europa.eu/digital-single-market/en/news/ehealth-action-plan-2012-2020-innovative-healthcare-
21st-century
86
https://www.covingtondigitalhealth.com/2018/05/summary-of-the-european-commissions-ehealth-strategy/
87
Professeur Hervé Dumez, Professeur Etienne Minvielle, Madame Laurie Marrauld, État des lieux de l’innovation
en santé numérique, November 2015
88
Topol E., The Creative Destruction of Medicine: How the Digital Revolution Will Create Better Health Care,
Basic Books, August 2013, p. 336

Market study on telemedicine
Final Report
94
between data security and data sharing. Addressing aspects of privacy, confidentiality,
and data security is vital in order to give new impetus to telemedicine.
Liability issues
Legislations are not clear regarding liability and accountability of practitioners in
telemedicine. Therefore, physicians are reluctant to embrace telemedicine since they are
worried about being made responsible for failing to act.
For instance, a telemonitoring device such as LifeWatch transmits wirelessly and
continuously asymptotic and symptomatic arrhythmia to clinicians. If the doctor is busy
and does not have time to review the patient’s daily activity, he/she might not notice the
patient’s condition worsening. If the patient passes away, would the doctor be made
responsible for held liable?
The legal framework in EU countries does not provide explicit rules about liability. In
the Netherlands, for instance, due to the lack of regulation, healthcare professionals are
fearful of using telemedicine because they deem that the nature of remote or virtual care
exposes them to the risk of malpractice.
Challenge of medical licensure or credentialing
Especially in the US, healthcare providers must have a medical license available in the
patient’s state to deliver care. This regulation acts a barrier to telemedicine expansion
within the country.
Nonetheless, progress has been made. The Interstate Medical Licensure Compact,
supported by the American Medical Association and enforced in 2015 by 17 states,
allowed physicians to practice telemedicine in these states.
In the EU, each country has its own rules for delivering a medical license to practice
healthcare in its territory. Although the procedure to get a license in a Member State is
generally smoothed for doctors coming from EU countries, the heterogeneity of the
models makes it difficult for professionals to understand which conditions they must meet
to practice in a specific country.
3.1.3. Social security conditions
Telemedicine reimbursement rules within the country
In almost all countries, reimbursement schemes of teleconsultation or other telemedicine
services to patients remain vague or non-existent. Health funds are often held
responsible for narrow restrictions on the coverage and reimbursement of
telemedicine services. The slow pace of legislation change is not helping solve this
problem.
Sometimes, market players have reached agreements with insurance companies
(Germany, the Netherlands) or public healthcare providers (the UK, Italy, Spain) but they
are the results of time- and resource-consuming negotiations. While some
telemedicine services are now eligible for reimbursement, patients still bear the cost of
most of them. In addition, reimbursement from health funds often takes place if specific
conditions are met (e.g. service provided in a doctor’s office or patient living in a rural
area). Thus, non-transparent and complex reimbursement models confuse patients
who are not able to understand which services are reimbursable and to what extent.

Market study on telemedicine
Final Report
95
For instance, in Italy, the outcome of the cooperation between the government and the
regions was the integration of telemedicine into the definitions of home hospitalisation
(OAD) and integrated home care (ADI). However, these efforts have been fruitless since
the reimbursement schemes and financing structures have not kept pace with these
changes. Misalignment between policy and execution accounted for the hardship of
generalising telemedicine practice in the case of MyDoctor@home for example.
Without proper reimbursement rules, the telemedicine market will not grow as expected.
France recently decided to reimburse teleconsultation exactly as if it was a face-to-face
consultation. This rule starts on Sept 2018. It will be interesting to monitor the impact on
the development of teleconsultations in the short and long term.
Telemedicine reimbursement rules between EU countries
This reimbursement issue is even more blatant between EU countries. Since
reimbursement schemes vary from state to state, patients struggle to be reimbursed for
telemedicine services provided abroad.
Directive 2011/24/EU on patients’ right in cross-border healthcare defines the conditions
under which a patient may receive medical care and reimbursement from another EU
country. Furthermore, Coordination regulations (EC) Nos. 883/04 and 987/09 entitle
insured individuals to receive healthcare elsewhere within the EU or EEA and in
Switzerland. It covers healthcare costs, as well as the prescription and delivery of
medications and medical devices as it would in their home country. Usually, patients pay
upfront and get reimbursed afterwards by their home health fund on the same amount
they would have received in their own country (article 7).
However, the rules on reimbursement are valid only if the treatment is available and
covered in the patient’s home country. These rules are obviously applicable to
telemedicine but national legal frameworks and reimbursement schemes (especially for
telemedicine) are unclear and highly heterogeneous. National health funds might not
reimburse the same (tele)medical acts and if they do, not necessarily in the same amount
or proportion. Besides, a health fund can refuse to reimburse a patient if it deems that
the medical treatment could have been delivered in the home country.

Market study on telemedicine
Final Report
96
3.1.4. Industrial and technical conditions
Figure 44: Industrial and technical barriers
Lack of infrastructure
EU countries are also facing a lack of IT infrastructure, which is generally too basic or
inadequate to integrate the telemedicine solutions currently available.
Insufficient funding and poor investment in modern equipment for hospitals do not permit
to bridge the gap between the innovative and advanced telemedicine products/services
and the existing infrastructure. Yet, setting up reliable and broadband systems is crucial
to provide quality telemedicine practices. For instance, the lagging information and
process management infrastructure in Ireland is hampering the widespread adoption of
telemedicine in the country
89
.
In addition, countries are struggling to implement interconnected networks across various
levels of the health sector. A uniform and standardised IT infrastructure would
provide network synergies and bring significant improvements in information
and resource flows. Instead, the heterogeneity of the systems adopted by the entities
in the health sector results in time and resource being wasted.
In Slovenia, the government’s failure to reach its ambitious telemedicine goals (defined in
2005 by the Ministry of Health) results in persistent bureaucratic procedures and non-
functional IT infrastructure in the healthcare sector
90
.
Lack of IT training/personnel
Another identified barrier to the uptake of telemedicine is the lack of IT training for
healthcare providers. Physicians in general are not familiar with the new technologies and
need some professional IT support to understand how to properly use them in a medical
context. Telemedicine is constantly developing, market players are flooding the market
with new solutions and thus, doctors are struggling to keep pace with the latest
developments.
Telemedicine technologies often require highly specialised knowledge. Yet, there is an
inadequate pool of specialised medical personnel capable of exploiting them since
89
Department of Health, eHealth strategy for Ireland, 2014
90
Stanimirović D., Mirko Vintar M., Analysis Of E-Health Development In Slovenia, 2013

Market study on telemedicine
Final Report
97
few physicians have the necessary skills to introduce technology in their patients’
pathway.
A reason for this is that practical training in telemedicine has not been firmly
incorporated into continuing education. Both at EU and national level, updated
training programmes must be created to increase digital literacy and to expand the
workforce’s capabilities regarding the use of technology in telemedicine.
It is fundamental to recall that telemedicine means introducing ICT to help improve
healthcare delivery and is not meant to replace healthcare providers. Therefore,
authorities should not overlook investments in human capital, which are essential for
effective implementation of the technology.
Less technologically advanced countries
Some EU countries are lagging behind regarding technological development. Limited
internet connectivity in medical establishments combined with the population’s
poor computer literacy undermines the potential of telemedicine deployment in those
countries.
In some countries such as Ireland, physicians also reported system crashes linked to
power outages, computer viruses or hardware failures. Having an IT infrastructure in
medical establishments is a first step but it needs to be efficient and reliable to
support leading-edge technologies.
A number of less developed EU countries still rely on “paper-based” hospitals in which the
telemedicine potential is very restricted. Not only would digital procedures mean
increased workload for healthcare professionals because these procedures would
coexist with the usual paperwork but they would increase the risk of information and
data misalignment. Missing or outdated technology represents an additional barrier to
telemedicine adoption.
3.1.5. Knowledge conditions
Lack of evidence, awareness, education
The lack of unambiguous evidence of the benefits holds up widespread adoption of
telemedicine by all stakeholders. A stronger consensus on cost-effectiveness would
enable patients, healthcare professionals, insurance companies and policymakers to
understand the potential of telemedicine in healthcare. However, EU citizens are not fully
aware of it.
Scientific-based evidence of telemedicine benefits is not abundant. Besides,
scientific papers proving the cost-effectiveness of telemedicine solutions generally use a
jargon inaccessible to non-experts. Consequently, policy-makers are unwilling to make
hasty decisions on legislation, reimbursement and funding as long as there is little
available and standardised evidence. This in turn feeds through to market players who
cannot spread out their products and services and have to keep prices high due to
missing market scales.
Higher education ability to deliver a skilled workforce
Medical schools have not yet firmly incorporated telemedicine training into their
academic standards. Yet, it would be wise to teach students the difference between
remote care and in-person care as well as the benefits, limitations and regulations of

Market study on telemedicine
Final Report
98
telemedicine. It would familiarise them with telemedicine practices and enable them to
integrate them properly in care delivery.
Germany has started to address this issue and the University Medical Centre Mainz offers
a teaching encompassing five modules, which aim at upgrading students’ skills in
telemedicine. This makes sense since today’s medical students have grown up using
digital technology and thus are much more comfortable with integrating it in the
delivery of medical care. This high digital literacy must nevertheless be sustained by
formal and structured training in order to ensure that they will provide high-quality
telemedical care.
3.1.6. Financial conditions
Funding matters
Telemedicine uptake also relies on the financial resources made available, essentially to
cover for technology purchase upfront costs. The question is who should be
responsible for the funding.
The studies conducted by JRC showed that, regardless of the source, funding is crucial
to incentivise telemedicine initiatives.
However, funding relying only on the industry is not viable. Funding must also come from
national, regional or EC budgets to be sustainable. It constitutes a prominent driver for
SME and start-up involvement since it enables them to benefit from subsidies or
preferential loans from the government.
For instance, in France, since 2012, the Fonds d’Intervention Régional (FIR) has invested
€40 million each year in actions and experiments validated by the regional health
agencies (ARS) in order to stimulate telemedicine projects and foster innovation. Yet less
than 50% of this annual budget has been effectively dedicated to telemedicine, the
regional health agencies using the “fungibility principle” to allocate these resources to
other projects. In this case, the initial objective of promoting telemedicine initiatives is
partially compromised by the poor management of resources. Although it needs to
further efforts, the UK government also provides funding through the Technology
Strategy Board (TSB) or the Department of Health. The TSB has notably supported
services and applications for the elderly such as the ALIP platform, Year Zero or Living It
Up.
EU countries all have different funding patterns. For France and the UK, the
government might be a key player but other countries just rely heavily on European
Commission funding. This happens when national or regional sources are quite limited
and when governments cannot afford to invest in telemedicine. The European
Commission has implemented many EU-funded projects (MOMENTUM, United4Health and
Renewing Health for instance): the subsidies granted, however, must supplement other
funding since the EU will not fund 100% of a project. Therefore, EU countries need to
boost co-payment for telemedicine projects or solutions by strengthening
industry commitment in telemedicine funding. To drive companies to invest in
telemedicine projects, governments must create research incentives and tax benefits to
get the industry involved in this sector.
Nevertheless, some EU countries act as role models in terms of funding. In Denmark, the
PWT Foundation is providing many pilot projects with substantial resources: it endeavours

Market study on telemedicine
Final Report
99
to narrow the gap between the R&D phase and the implementation and distribution
phases. As of 2010, 54 local demonstration projects had received funding from the PWT
Foundation. In Scotland, success stories in funding to overcome financial barriers also
exist. The Scottish government’s budget for eHealth was €112.5 million in 2012, of which
the great majority goes to NHS National Services Scotland to fund national eHealth
projects. For instance, the TeleScot project, a program of academic research investigating
telemetric-supported telemonitoring of a number of long-term health conditions, received
funding of around €2.375 million from different sources.
3.1.7. Market conditions
Fragmentation between primary and secondary care
The fragmentation between primary and secondary health care is also slowing down the
adoption of telemedicine solutions.
Professionals in hospitals believe that the initial impetus must come from GPs
because they are in a position to identify and convince the patients more suited to use
telemedicine in their care pathway. This first approach from GPs could help hospitals to
resort to these technologies when they face full bed occupancy and must discharge some
of their patients early.
Nonetheless, there is a lack of coordination between primary and secondary care
professionals, who shift the blame onto each other regarding the slow deployment of
telemedicine. In addition, the lack of incentives and of e-readiness is preventing GPs from
embracing telemedicine: this results in a stalemate within the medical sector.
Fragmentation of the solutions
The solutions developed by the various companies are usually not interoperable in
relation to how data is structured, stored, transmitted and accessed. They are
therefore deeply fragmented instead of being integrated. As a consequence, the
telemedicine solutions implemented in a country’s hospitals or regions might be
completely ineffective elsewhere.
This problem underlines how critical it is to adopt common standards for
telemedicine. Interoperability is key to efficiently integrating the new solutions into the
established systems.
Multilevel policy intervention and the involvement from all stakeholders, including the
industry, are required to improve this situation.
Buyers' fragmentation and constraints on market scale
The procurement process of telemedicine solutions differs from a country to another.
German companies negotiate with health funds whereas in Spain, companies have talks
with local healthcare providers. In Italy, they first need to approach regions and then
municipalities.
These heterogeneous models add confusion for companies that would like to make
their solutions available internationally since they do not necessarily know who they
should approach to do so. Companies struggling to enter foreign markets do not benefit
from economies of scale and thus keep a strong “home base”.

Market study on telemedicine
Final Report
100
In conclusion, the fragmentation of solutions, of buyers and between primary and
secondary care acts as a heavy barrier to telemedicine and seriously restricts the
potential for its widespread adoption across EU countries.

Market study on telemedicine
Final Report
101
3.2. SWOT analysis of the telemedicine framework
Based on the review of literature, we provide the following SWOT analysis
91
,
92
,
93
,
94
,
95
:
Table 2: SWOT analysis of the telemedicine framework
91
SIMPHS 2 (2012), JRC
92
Hoerbst A, Schweitzer M. A systematic investigation on barriers and critical success factors for Clinical
Information Systems in integrated care settings.
93
Study on Big Data in Public Health, Telemedicine and Healthcare, Final Report, December 2016
94
eHealth in Europe - Status and Challenges
95
Report on the public consultation on eHealth Action Plan 2012-2020
Strengths
Weaknesses
Cultural conditions
- Overall high digital literacy of the population.
Regulatory and policy conditions
- National policy/strategy in telemedicine.
- Policy focus on chronic disease management.
Industrial/technical conditions
- Multiplication of innovative and advanced solutions.
Financial conditions
- Dedicated budget from the EC to telemedicine
projects.
- National or regional funding mechanisms promoting
sustainability of initiatives.
Market conditions
- Cost-effectiveness of telemedicine solutions.
Cultural conditions
- Lack of patient/social awareness of telemedicine.
- Fear of malpractice among healthcare providers.
Regulatory and policy conditions
- Legal loopholes regarding liability and data
confidentiality and security.
- Poor regulatory framework, lack of standards and
guidelines.
- Misalignment of national policies might jeopardise an
EU-wide uniform approach to telemedicine.
- Different data privacy policies.
Social security conditions
- No clear and efficient reimbursement models.
Industrial/technical conditions
- Insufficient interoperability.
- Poor system reliability and response time.
Knowledge conditions
- Lack of scientific-based evidence of the benefits of
telemedicine.
- Shortage of trained staff in telemedicine.
Financial conditions
- Limited support from government.
Market conditions
- Complexity of relationship and interest management
between the various players and stakeholders.
- Interoperability challenges due to EU fragmentation.

Market study on telemedicine
Final Report
102
Opportunities
Threats
Cultural conditions
- Develop proper dissemination and communication
strategies to overcome GPs' reluctance.
- New generations more comfortable with using
technologies in healthcare.
- Use already interested public and private
stakeholders as levers to increase acceptance.
- Communication with the public can increase
awareness of how important open data and data
sharing are.
Regulatory and policy conditions
- New legislation can be the foundation of wider use of
telemedicine.
- Find common ground between Member States'
legislations and national standards.
- Define clear rules on liability when using
telemedicine solutions.
Social security conditions
- Develop of new reimbursement frameworks.
Industrial/technical conditions
- Technology needs to be flexible to meet users' needs
properly. It has to allow for personalisation (e.g.
parametrisation).
- Promote training of healthcare professionals and
end-users.
- Invest in IT infrastructure in hospitals or other
specialist facilities.
- Further cooperation with industry to ensure
interoperability and alignment with clinical protocols.
Knowledge conditions
- Achieving a minimum level of cross-linked
knowledge of all involved parties may facilitate wider
use of telemedicine solutions.
- Increased motivation for education and training in
telemedicine.
- Multiply health technology assessments (HTAs) to
obtain a systematic evaluation of properties, effects
and impacts of telemedicine.
- Greater involvement of HTA bodies recently that are
able to provide scientific-based evidence.
Financial conditions
- Diversify funding schemes and increase commitment
from the industry.
- Promote multi-source financing and public-private
partnerships in funding.
- Review existing incentives.
Cultural conditions
- Loss of the doctor-patient relationship and of the
social link.
- The elderly's resistance of technology in the care
process.
- Lack of experts' commitment to telemedicine
practices.
Regulatory and policy conditions
- Persisting lack of interoperability between solutions
and difficulty in aligning national standards and
protocols.
- Different political priorities and interests hindering
the wider use of telemedicine.
- Restrictive (privacy) laws might hinder data sharing.
Social security conditions
- Lack of coordination between EU countries in
establishing reimbursement rules.
Industrial/technical conditions
- Risk of technological flaws.
- Risk of data leaks.
- Data overload can create resistance.
- Different technological levels and advancement of
involved national bodies and stakeholders.
Knowledge conditions
- Dearth of impact assessments to balance the need
for sound evidence of telemedicine benefits. Reports
including qualitative and quantitative elements are
crucial to facilitate decision-making.
- The swift pace of technological change could cause
educational programmes to be outdated by the time
of their implementation.
Financial conditions
- Financial burden of initial investment in
telemedicine.
- Expensive solutions from some market players.
Market conditions
- Increased workload for healthcare professionals if
data coexists with paper.
- “Silo thinking” and lack of cooperation between
primary and secondary care.
- Enduring strong national focus from telemedicine
market players.
- Market players fear a potential loss of intellectual
property.

Market study on telemedicine
Final Report
103
3.3. Policy recommendations
3.3.1. Actions to overcome cultural barriers
Trigger pressure from the demand side
Patients from advanced countries in telemedicine expect GPs to be up to date
regarding ICT in healthcare. For instance, Danes consider doctors who are not
equipped with a PC during a consultation to be second-rate. Doctors are more likely to
adopt telemedicine and technologies in general when they are urged to do so by patients.
Launch appropriate communication campaigns
Another way to overcome this barrier is to set up suitable communication events to
raise awareness of telemedicine benefits. It is fundamental to emphasise that
technology is a complement rather than a substitute to face-to-face interaction. Dialogue
between all stakeholders (patients, healthcare professionals, market players, health
insurance companies) might be the best way to remove initial doubts and reservations
about telemedicine. In Italy, notable communication operations have been launched as
well as seminars to sensitise interested parties to the benefits of telemedicine.
Train healthcare professionals
Integrating technologies in doctors’ in-service training may release the full potential of
telemedicine development. It would enable the workforce to become more familiar
with telemedicine and thus to be more confident in using it in the care delivery.
In France, SF Telemed offers training in order to support all kinds of healthcare
professionals in their learning process of telemedicine. Similar training courses are
available in most EU countries but their prices are often prohibitive.
3.3.2. Actions to overcome regulatory barriers
Top-down approaches
Top-down approaches are useful levers to ensure interoperability and service quality
standards. The EU has a key role to play in this field. An effective policy would allow for
guidance, support and skills development likely to comply with central requirements,
regulations and incentives.
Countries such as Estonia have already tackled this interoperability issue. X-Road is an e-
solution that allows the nation’s various public and private sector databases to link up and
function in harmony
96
.
Government could establish funding eligibility criteria: projects that show significant
advances towards interoperability must receive assistance in priority.
96
https://e-estonia.com/solutions/interoperability-services/x-road/
Market conditions
- Leverage demand from patients in “ICT-advanced”
settings.
- Gain the support of GPs so that they can influence
their patients.

Market study on telemedicine
Final Report
104
Simplify and secure health data sharing
The Member States must allow effective collection, storage, processing and sharing of
health data and set up a clear data protection legal framework, with a simple and
workable patient consent procedure.
Decision makers need to implement these policies without jeopardising patients’
rights to privacy and confidentiality. Member States should set up governance
mechanisms to guarantee secure and fair use of data. The General Data Protection
Regulation (Regulation 2016/679) could be used as a model, as one of its objectives is to
protect the rights of natural persons.
Coordination
EU countries should harmonise their legal frameworks in order to make solutions
compatible and to enable cross-border telemedicine practices.
They also need to agree on terminology and definitions to share the same
language and align already existing standards before incorporating them into the
national legislation. Besides, existing standards should be updated regularly for two main
reasons: to keep pace with the ever-changing technological environment and to avoid
overlaps between existing and new standards that might create confusion.
3.3.3. Actions to overcome social security barriers
Public authorities are starting to address these social security barriers. Although few
telemedicine practices are currently eligible for reimbursement, all Member States are
redoubling their efforts to extend the scope of telemedicine by gradually adding
new acts covered by social security. Amplifying and simplifying reimbursement rules
could bring down major barriers and speed up telemedicine adoption.
In France, thanks to an initiative from the Health Ministry, representative unions of
private physicians and the health insurance fund have sat down together and opened
negotiations to extend reimbursement schemes in telemedicine, in particular for
teleconsultation. Articles 54 and 55 of the draft law on the financing of the health fund
(PLFSS 2018) set up the conditions to facilitate the eligibility and the registration
procedure for the reimbursement of telemedicine acts.
3.3.4. Actions to overcome industrial/technical barriers
Several avenues of thought to overcome industrial/technical barriers:
- Increase public investment to enhance the IT infrastructure and reinforce its
capacity to process information flows.
- Strengthen human capital so that the medical workforce meets the current
telemedicine requirements.
- Integrate practical training in the curricula of medical schools to ensure an
acceptable degree of technological skills development.
- The European Commission could fund appropriate initiatives aimed at facilitating
knowledge sharing.

Market study on telemedicine
Final Report
105
3.3.5. Actions to overcome knowledge barriers
In order to tackle knowledge barriers, EU countries need to promote scientific-based
evidence of telemedicine benefits in a language, which is accessible to any reader. A
number of patients and doctors are still reluctant to use telemedicine solutions because
they consider that there is still a lack of information as to their effectiveness. The main
findings of telemedicine studies should be made available through the right
communication channels to reach the entire population.
In a similar vein, telemedicine market players also need to learn how to correctly
market their service to patients because too many patients are not aware of the
existence of solutions that could suit their needs. Appropriate and targeted marketing
campaigns could be a good means for companies to make themselves more visible.
Finally, governments could fund additional training courses in telemedicine for GPs who
are the main contact points for patients and thus are able to convince the latter to use
telemedicine solutions to treat their diseases. Upskilling health professionals
(including medical doctors) in digital technologies is key for the digital transformation
of health.
3.3.6. Actions to overcome financial barriers
To get healthcare professionals involved in the development of telemedicine, EU countries
could implement pay-for-performance (P4P) schemes to reward doctors (bonuses, add-
on payments) when they meet patient satisfaction benchmarks in telemedicine.
The EC also has a role to play to remove financial barriers. It should steer suitable
investment to relevant initiatives in order to guarantee cost-effectiveness and
sustainability. However, the EC cannot be the only one responsible for funding. Member
States need to favour multi-source financing and public-private partnerships to
spread the initial financial burden and the risk of investment associated with the
development of new telemedicine solutions.
Another strategy to ensure interoperability is to make it a criterion in public procurement.
By setting interoperability standards that solutions should meet in order to be selected
during a public procurement process the EU and MS governments can stimulate
interoperability.
3.3.7. Actions to overcome market barriers
Recommendations for market players
97
:
- Examine the policy context and identify potential blockers and enablers. Market
players should develop their solutions around the enablers, and define workarounds
to resolve blocking elements. This includes mapping existing resources in relation to
the implementation context, financing, internet access, legislations, etc.
97
W. Gaafmans, F. Abadie, IPTS, Information Society Unit, eHealth team., SIMPHS 3, Guidelines for ICT-
supported Integrated Care, 19 May 2015.

Market study on telemedicine
Final Report
106
- Make sure that their telemedicine solutions meet defined user needs, focusing on
care professionals, patients or other key stakeholders. In addition, design and plan
awareness actions to make sure target users are informed about the benefits of the
new system.
Recommendations for governments:
- Appoint champions to advocate the initiative, monitor change management and
define new roles for care professionals whenever required, to implement the change
and coordinate the new care processes. “Innovators” and “early adopters” can foster
the use of innovative Big Data analytics.

Market study on telemedicine
Final Report
107
4. ECONOMIC ANALYSIS
Key takeaways
In essence, telemedicine is generally perceived and judged to be cost-effective in
73.3% of the cases addressed by the literature.
Neutral effects were discussed in 21.3% of the selected references, mainly in
systematic reviews.
Negative effects account for 5.6% of the studies.
Further adoption of telemedicine increases benefits: it reduces costs (consultation
costs, travel costs, time spend) and increases patient survival and life quality.
To overcome the barriers there is a need for more scientific evidence for its
efficiency and large scale experiments to assess the impact of a wider deployment.
Raising awareness (patients, doctors), stimulate integration between stakeholders
and reimbursement are keys to success.
The present chapter develops an economic assessment framework with the objective of
evaluating the potential benefits of future deployment of telemedicine tools and services
across the EU. This assessment is relies on the development of an economic decision
model
98
based on insights drawn from scientific research.
This economic model enables a cost-effectiveness analysis (CEA) of the future EU-wide
deployment of telemedicine solutions under different scenarios; for the sake of
parsimony, we analyse two specific cases of potential future deployment: a) promoting
telemedicine for any type of disease, and b) promoting the use of telemedicine for major
chronic diseases. For each of these two cases, we and investigate the implications of a
"baseline" scenario (business as usual) and an alternative scenario (efforts to increase the
use of telemedicine tools and services).
The development of this assessment framework is based on a two step approach; the
first step involves the research design and collection of information on the cost-
effectiveness of telemedicine solutions and services adoption. The activities undertaken in
during this step are the literature review on the cost-effectiveness of telemedicine
solutions. The second step of the approach involves the development of the economic
model itself, the activities performed during this step include the implementation of the
decision model, and the estimation of the total costs and benefits associated to
each scenario under each particular case of future deployment.
The key objectives addressed by the activities undertaken within the scope of the present
chapter are to highlight the evidence and model the cost-effectiveness in using
telemedicine to promote health, minimise illness and disability, and generally improve
quality of live and longevity.
98
Morgan, S., et al. (2007). Assessing the value of medical devices. University of Nottingham – Brunel
University. Discussion document

Market study on telemedicine
Final Report
108
4.1. Systematic review of cost-effectiveness studies and data collection
The systematic review of cost-effectiveness studies carried out in this section aims at
providing an initial insight into the capacity of telemedicine solutions to improve the
quality of healthcare services and reduce the costs incurred, in comparison with
traditional care. This assessment made it possible to distinguish positive effects in terms
of cost-efficiency, from neutral and negative effects raised in the health economics
literature.
A five-stage scoping review methodology was implemented to identify and analyse the
economic literature on the cost efficiency of telemedicine through the following steps:
1. Identification of the literature sources and repositories in order to ensure access to
a relatively wide sample of articles and reports;
2. Identification of relevant studies by interrogating the repositories on several key
topics (cost-efficiency and telemedicine, eHealth, mHealth or digital health);
3. Selection of relevant studies for review;
4. Creation of a database from the selected literature for analysis, and
5. Appraisal and analysis of the literature.
The data collection took place between October and November 2017, focusing on
electronic searches conducted across several databases referenced in the Tender
Specifications for the study and corresponding proposal; namely the Cochrane Database
of Systematic Reviews and the Database of Abstracts of Reviews of Effectiveness (DARE),
the CRD database of the University of New York, Jstor, Science Direct, and PubMed.
The output of the literature review and research design comprises a database of a total of
190 scientific references whose appraisal was performed on the information present in
their abstract, their characteristics on the topic, and information about the study. A
PRISMA assessment of the criteria led to the following outcome:
44 articles not fit for analysis, as the topic did not match the needs of this study;
61 articles fit for analysis;
28 articles presenting a systematic review on cost-effectiveness;
47 articles addressing seldom benefits or barriers to adoption of telemedicine services
without reaching a conclusion on their cost-efficiency.
For the purpose of this analysis, we used the subset of 89 literature references (61
studies and 28 reviews) addressing the cost-efficiency of telemedicine solutions to
perform a second appraisal, this time indicating the conclusions obtained.

Market study on telemedicine
Final Report
109
Figure 45 : Screening flow diagram of included literature
The results from this review show that in essence, telemedicine is generally perceived
and judged to be cost-effective in 73.3% of the cases addressed by the
literature while neutral effects were discussed in 21.3% of the selected references. The
latter mainly found in systematic reviews that reach a conclusion on the lack of robust
and high quality studies on the evaluation of telemedicine services, advocate for further
research and evaluation, and do not pronounce in favour of a positive or negative
assessment. The following Figure provides an illustration of the results of the sentiment
analysis.
Figure 46: Distribution of cost-effective effects from the assessed literature
Furthermore, the studies under assessment were categorised according to a broad
classification of the type of telemedicine solution addressed; this classification
comprises tele-expertise (interaction only between healthcare professionals),
IdentificationScreeningEligibilityIncluded
Records identified
through database
searching
(n = 191)
Records screened on the
basis of their abstract
(n = 191)
Records excluded
(n = 44)
Records assessed for
eligibility
(n = 136)
Articles addressing
benefits and barriers of
telemedicine without
discussing cost
effectiveness
(n = 47)
Original research articles
discussing cost
effectiveness and included
in quantitative and
qualitative synthesis
(n = 61)
Systematic reviews
discussing cost
effectiveness and included
in quantitative and
qualitative synthesis
(n = 28)
Positive
73,03%
Neutral
21,35%
Negative
5,62%

Market study on telemedicine
Final Report
110
teleconsultation (e.g. selection, diagnosis, and treatment of patients), and telemonitoring
(e.g. follow-up treatment, telecare, self-management). The distribution of these studies
indicates that the majority of solutions are represented by teleconsultation solutions
(38.2% share) and telemonitoring solutions (43.8% share) or programs discussed within
the sample of literature references. Finally, a 16.8% share of the sample, mainly
represented by systematic reviews, discussed either the two main categories
(teleconsultation and telemonitoring) or all categories including tele-expertise.
Figure 47: Distribution of telemedicine solutions in the assessed literature
In addition to these descriptive analyses of the study assessment, we focused on the two
principal categories discussed in the literature, teleconsultation and telemonitoring
99
,
to better understand their cost-effectiveness and potential differences, as indicated in the
conclusions from the assessment of studies. This analysis shows that in general both
types of telemedicine solutions are assessed as cost-effective in the literature in a
relatively similar proportion, with higher shares in telemonitoring due to the higher
concentration of the selected literature on this type of telemedicine solutions. This
difference is mainly driven by the neutral conclusions on cost-effectiveness, which are
twice as high in teleconsultation as in telemonitoring. These observations and the
conclusions observed in, and drawn from the sample of studies under assessment, may
point to a longer period of adoption or to major barriers to adoption and scale in the case
of teleconsultation solutions. The contingency Table below provides details on this
distribution.
99
Which matches with the findings of the solution mapping exercise where telemonitoring is the predominant
type of intervention for telemedicine solutions
Teleconsultation
38,20%
Telemonitoring
43,82%
Teleexpertise
1,12%
Several
16,85%

Market study on telemedicine
Final Report
111
Table 3: Contingency table on the distribution of types of telemedicine solution and associated cost-
efficiency
Telemedicine\Cost-
effectiveness
Positive
Negative
Neutral
Total
Teleconsultation
31.5%
2.2%
4.5%
38.2%
Telemonitoring
34.8%
3.4%
5.6%
43.8%
Teleexpertise
0.0%
0.0%
1.1%
1.1%
Several
6.7%
0.0%
10.1%
16.9%
Total
73.0%
5.6%
21.3%
100.0%
4.2. Cost-effectiveness of telemedicine solutions
The literature review on the cost effectiveness of telemedicine solutions and services led
to the collection of data related to monetary costs and benefits (in terms of costs, time
and logistics savings, as well as improvement of key performance indicators such as
morbidity and quality adjusted life years).
The collected information was used to feed the economic models that allows to evaluate
the potential effectiveness of future development. The two models (any disease, or
chronic diseases only) were evaluated under two different scenarios for comparison.
Baseline scenarios denote no intervention and business as usual. Alternative
scenarios denote an increased rate of adoption of telemedicine.
4.2.1. Baseline scenario – No EU driven efforts to adopt of telemedicine
solutions
The baseline scenario represents the current situation on the European telemedicine
market; it used as a point of reference for the assessment of the EU wide deployment of
telemedicine solutions. Under this scenario, we consider two models, one based on the
total population of the EEA area, and the second focused on the chronically ill population.
Under this scenario, even though society, healthcare providers and decision makers know
that we need to go through a change of paradigm, decisions concerning the business
models to be adopted and actions in favour of the uptake are still hardly taken or difficult
to implement.
There is an unclear vision on the utility of telemedicine pointing out to its use to either
increase performance (by treating more individuals with less resources), or its use for the
provision of optimal care (by increasing its quality and efficiency). Regulators still try to
understand better how to manipulate the levers to trigger a wide adoption and
implementation of telemedicine.
In this context, the regulator needs to set clear rules in terms of security, confidentiality,
and ethics. It is important to understand that the use of telemedicine tools and services
require the convergence of different professions, types of organizations and technical
infrastructure is often differ and fail to integrate together. This environment creates
certain distrust and defiance from the medical professions to the technology itself.

Market study on telemedicine
Final Report
112
In addition, the human factor is still very important in the healthcare area; patients will
only change slowly. However, today patients have started modifying their behaviour, they
usually turn first to the internet for comparison of symptoms or plain information, and
then consult the doctor with a preconceived idea on their health status, that may or may
not be correct. Changes are starting to take place, although very slowly.
Market characteristics
Under the baseline scenario, without any EU intervention, patterns in the demand for
telemedicine solutions will not be affected by the sponsoring effects of the integration of
telemedicine within national health systems, or by the harmonisation of standards,
regulation, and security requirements. In essence, then demand will not be aggregated at
national levels, and therefore no-synergies between private and institutional users, but
also between EU MSs will be achieved.
On the supply side, the market for EU public service users will remain highly fragmented,
with telemedicine, e-health and m-health market players segmenting, and mainly
investing on more profitable markets defined by catalysing factors for the adoption of
such technologies, e.g. demographics, health infrastructure, reimbursement systems, etc.
On the governmental side, national health systems would need to be challenged at some
stage regarding the opportunity cost and cost-effectiveness of not- adopting telemedicine
solutions at large scale, without any guarantee of meeting the increasing service capacity
needed to meet the needs of a growing population.
The demand for telemedicine solutions will thus keep growing over time for as long as the
population dynamics, the investment in health infrastructure, and capital and operational
expenditure in human resources create the appropriate environment for endogenous
growth.
Scenario outlook
Currently, business models behind national health systems differ in many points; for
instance, while in some countries the aim is to keep patients away from care services by
means of prevention and appropriate care, in other countries the aim is to provide them
with more services. In addition, depending on how care services are financed, through
insurance or social welfare the optimal business models for the provision of telemedicine
will completely differ across the EU territories. Finally, it is worth noticing that the more
patients there are, the higher the burden on health systems will be due to more
procedures and complexity. Thus, more expenditure will be required from the society. It
is also necessary to note, that are the baby boom generation is getting old, the national
health systems will be in pressure to provide more services, especially geriatric services.
In several EU health systems, the link between how the system is financed and the need
to ensure a revenue from patients implies a business model where health services are
pushed to patients therefore creating a barrier for the uptake of telemedicine directly
related to the adopted reimbursement model.
Under this scenario, care services are undergoing a transformation process driven not
only by technology, but also by the need to cover many more patients as the EU
demographic trend increases and a larger share of individuals demand not only base care
but also higher quality care services.
The capacity reduction of healthcare providers is illustrated in the development of
infrastructure (i.e. hospitals) expected to serve twice as many patients with less

Market study on telemedicine
Final Report
113
resources; this dynamic implies the need to find solutions to provide the same level of
services to patients with lower capacity, therefore aiming for the uptake of telemedicine.
These solutions need to transform health services and procedures to be more efficient.
This is a change in paradigm from a physical presence at the hospital to a technology-
based alternative; in the coming years, it will be difficult for healthcare providers in the
EU to survive unless they move into that alternative.
Need for evidence and large demonstrators
In order to allow for a natural uptake of telemedicine without the support of coordinated
efforts of Member States, a base of clinical evidence is required to demonstrate the actual
benefits from adoption. This evidence base is costly and time consuming, and it becomes
more and more clear that we are overselling the economic potential of telemedicine
without actual evidence. Telemedicine market players are away from good evidence about
the efficiency of the service delivery hence failing to convince at a large scale. Even if a
lot has been done in terms of specific effectiveness studies in the recent years, the issue
is rather about the lack of large-scale implementation and the expected benefits. The
literature review on telemedicine cost-effectiveness allows observing many specific
studies but lack large-scale demonstrators. Efforts and investments are needed to build
good evidence, as it is difficult to implement large-scale solutions unless there is proof
about the benefits.
Moreover, it is important to invest in solutions that are easily integrated into the daily
operational systems of healthcare. Clinicians should be able to work in the systems that
they are used to regardless of the interfaces they are using; today the integration is the
most important feature in the process of adoption, but is underestimated and overlooked
perhaps due to its complexity. In many cases across the EU, deployment initiatives have
lacked good integration. Probably the technical development will probably help trigger the
uptake of telemedicine, but the trickiest part will be to change the organisations, the
procedures, and the knowledge embedded in the personnel.
Often, barriers are placed on testing pilot deployment initiatives. It is difficult to convince
the developers to invest heavily in the integration of the technology in the pilot since the
risk of failure is highly expensive, while on the other hand if the integration is not
optimal, then the likelihood of failure is important since the pilot would not be able to
deliver the expected outcome.
Enabling diffusion and adoption of telemedicine solutions across society
Further adoption of telemedicine under this scenario will require the development of
appropriate frameworks to deal with patient’s information, its storage, and is permissions
of access. The regulator needs to provide clear rules on the rights of the patients and
relatives to access and grant permissions to patient data; in essence, the problem is that
patients should have the right to decide about the use of their data, but healthcare
providers need to be granted access to this data thoroughly.
In addition, the main role of the regulator is therefore to provide clear data utilisation
standards as today the telemedicine environment is yet very wild, with many actors start
operating in this area for business opportunities, making it hard to assess the quality of

Market study on telemedicine
Final Report
114
their offers for professional and end users. Telemedicine should allow to access data and
clear regulations for accessing patient data. While health professionals should be able to
access patient data thoroughly, they should also have appropriate training and
certification by independent bodies to ensure that patient data is not misused or handed
to third parties. On the other hand, the patient, as a citizen, has the right to decide what
to disclose on not, and be reassured about data privacy and security measures framing
the use of his data.
As an illustration, in the Netherlands, healthcare provision in 2020 will require by law the
disclosure of all medical data to the patient in a “personal health environment”. This
requirement will give the right to citizens to look at and store their health records, and
will thus imply a change in the behaviour of practitioners. Consequently, there is a need
for a centralized system that sets the rules on the utilization of a public utility, which
should not be in private hands. Citizens are entitled to the rights of their health data. In
essence, the main role for regulators is to protect the electronic health records of patients
to spur optimal healthcare systems aiming at delivering high quality healthcare with lower
resources.
4.2.2. Alternative scenario – EU deployment of telemedicine solutions
increase by 5% rate of adoption
The alternative scenario is defined by a market set up where demand and supply
dynamics are also affected by the sponsoring effects of a technology adoption by national
health systems. Under this scenario, we present two models, one based on the total
population of the EEA, and the other limiting the scope to the chronically ill population of
the EEA (suffering from asthma, COPD, diabetes and heart failure).
According to the first model, telemedicine deployment across the EEA National Health
Systems allows to aggregate part of the demand for telemedicine solutions for private
and institutional users. However, in this specific case, aggregation is performed
indistinctively of telemedicine market segments based on different diseases.
The necessary standards and regulatory requirements will also need to be developed, and
differentiated per family of telemedicine solutions. Under this scenario, future health
infrastructure investments will also be required to match the needs of a wide scale
telemedicine deployment, although the magnitude of such investments will be much
higher than those necessary under the previous scenario given the wider scope of
solutions included under the present scenario. As described previously, investments will
be made by both private companies and health institutions, when they see a viable
business case.
Under the second model, the focus is on the solutions for chronic diseases (i.e. asthma,
COPD, chronic heart disease, and diabetes) that would bring the most benefits from a
societal point of view, since these refer to older people for which remote medical care
improves their quality of life.
The necessary standards and regulatory requirements will need to be developed, probably
differentiated per family of telemedicine solutions specific to the management, treatment
and monitoring of a specific chronic disease. Under this scenario, future health
infrastructure investments and awareness efforts are necessary to match the needs of a

Market study on telemedicine
Final Report
115
wide scale telemedicine deployment. As these investments could be significant, the role of
the state is increased. However, a high initial investment will pay off in the longer term as
patients with chronic diseases cost heavily to the health insurance systems.
Since in many cases, the main telemedicine consumers are practitioners themselves
(communications between GPs and specialists), the uptake of telemedicine services and
tools under this scenario have the potential to increase the quality and speed of diagnosis
and treatment, and significantly improve the life style of individuals suffering from chronic
diseases.
Under this specific model, the focus on specific chronic diseases is due to the burden
these represent for national health systems and to the current dynamics of decreasing
amounts of the medical personnel relative to the increasing suffering population.
Market characteristics
Under this scenario, assuming there is consensus amongst Member States, telemedicine
deployment will rely on the harmonisation of standards and regulations for all the
commercial solutions available in this specific market segment, which can be identified by
national health systems.
Market players willing to take part in the deployment will then need to commit to meet a
certain demand and its dynamics in the short, mid, and long term to guarantee the
access to these solutions by national health systems. As an incentive to join the
deployment, telemedicine market players would receive a commitment (in terms of
demand) from the different national health systems. Such interactions between
commercial companies and healthcare institutions should be able to guarantee the
provision of telemedicine capacity and services at a low prices for the management,
treatment and monitoring of patients suffering from the specific chronic diseases selected
for coverage under the deployment initiative.
Scenario outlook
Under this scenario, where investments and efforts are made to partially deploy
telemedicine solutions across the EU by focusing on those addressing chronic diseases,
benefits for society can be considerable.
These benefits mainly relate to the prevention of productivity losses as people can reduce
the time for diagnosis and consultation. In addition, as patient management simplifies
thanks to the technology, it is expected that the medical personnel will focus on care
activities while reducing the burden of administrative activities. Further benefits for
society will include an increase in the base medical resources, knowledge, experience and
health that will be made accessible to a wider audience, rendering the patient
management and treatment process more fluid, making it more comfortable for
individuals to receive care.
The improvement of the quality of healthcare services with an increase in comfort for the
patient will be helpful in the reduction of costs because preventive medicine will be widely
accessible. This improvement process has the possibility to trigger a virtuous cycle of
better information and education of both the medical personnel and the patient.
Telemedicine can thus guide people and provide preventive medicine in order to reduce
national health expenditure. In addition, as telemedicine is increasingly adopted and
gains of knowledge and experience across different professions take place, standards and

Market study on telemedicine
Final Report
116
prevention will also be improved in a virtuous cycle. These increasing returns to adoption
will be the base of the future improvement of the healthcare system.
The provision of the infrastructure will be costly in the beginning of the deployment, but
in the middle term there are huge possibilities for cost saving and improvement in quality
of life. Overall telemedicine will be cost-effective for society, as the deployment
telemedicine across the EU will trigger scalability and increasing returns to adoption.
The main or leading market players in Europe will be those who have demonstrated the
utility of the technology through evidence. For instance, today, there are many examples
in home telemonitoring. These are evolving fast in the technical field, although it is
different on the side of the adoption by institutional users.
Indeed, the dynamics of the technology development is faster than the adjustment of
behaviour in the healthcare profession in general terms; in addition, since healthcare is
about information, support, fears, high expectations etc., and therefore the processes
used by the medical personnel evolve at a very slow pace. Furthermore, the lack of
awareness and integration issues between technologies and medical personnel can slow
down the dynamics of the institutional demand. There is therefore a need for eHealth
awareness campaigns at all levels, including trainings for young students in medical
professions who are not sufficiently aware of the technologies and the way the can be
used.
The demand under this scenario comes from the healthcare systems themselves who
need to rethink the how the care services they provide can be better coordinated to suit
the total population and its demographic characteristics. The way national health systems
take advantage of the enabling technologies in order to optimize their organizations will
determine the success of deployment and its related benefits.
In the short term, healthcare providers will need to demonstrate that the technology will
reduce relative costs and expenditure associated to any individual of the population. The
question is how to measure this cost-effectiveness at such a large scale. In consequence,
there is a need to develop better evaluation systems bases on scientific evidence in order
to convince the society to adopt and use telemedicine on a regular basis. It is a challenge
to ensure the continuity of healthcare while at the same time asking for performance
improvement.
Incentivising healthcare providers to adopt telemedicine solutions widely
Since most of the benefits and costs savings drawn from the uptake of telemedicine will
mostly address citizens and municipalities, different players, especially technology market
players will observe an uneven distribution of these benefits. It will become difficult to
incentives for investment in present of asymmetry of benefits between patients and
healthcare providers; indeed the ones biggest investments will be made by healthcare
providers, while the biggest benefits will go to citizens in general. In consequence, there
is a need to find new ways and incentives to support and deliver care services by
leveraging the use of technology to improve patient management and treatment.
Efforts from decision makers will thus be needed to enable ease and speed up the
activities of healthcare providers, pushing them rapidly towards saving on low-value time
and space. Since the biggest savings will be at the patient level, it will also be important
to highlight and better describe the benefits for healthcare providers.

Market study on telemedicine
Final Report
117
Raising awareness for a better management of the healthcare
Many awareness actions take place at the EU level, although not always focused on
chronic diseases. Evidence about the effectiveness of telemedicine is needed in these
cases to convince practitioners. Some countries are ahead in terms of support, adoption
and willingness such as Estonia and the Nordic countries. As soon as successful cases in a
country demonstrate the utility of telemedicine, practitioners accept it as valid and are
more likely to engage in the uptake process.
In addition, it is worth noticing that a better coordination of healthcare systems through
technology in the case of chronic diseases, in particular telemedicine tools and services
for recurrent consultation and prescription, expertise and monitoring will help address the
issues related to the appropriate care given to patients and prevent the waste in the
medical industry. This feature of telemedicine uptake is related to green care and the
capacity of technology to enable a better follow up of the patient and his needs, and limit
waste in terms of food, transport, emission, and overconsumption.
Demonstrating the utility of telemedicine solutions across society
Considering the fact that since technology advances quite rapidly, tools are often very
advanced but fail to provide evidence of their utility and efficiency. Providers will need to
demonstrate the effectiveness of the technology, which as we have discussed in the
previous sections, lacks from evidence about its effectiveness, therefore slowing down the
telemedicine adoption process. It becomes necessary to better understand whether there
may be adverse effects of an increased use of telemedicine, especially when considering a
full deployment; for instance, tele-monitoring can easily lead to increased medication and
dependence as highlighted by a few studies.
Their main role refers to normalization and standardization. The issue of a reliable
exchange of patient information for different means (better diagnosis, patient
management, follow-up through telemedicine) is highly political.
4.2.3. Description of variables used for economic analysis
The economic analysis implemented to estimate the costs and benefits associated to each
of the scenarios under study relies on a series of parameters gathered from the literature
review, as well as studies on the adoption of remote medical technologies and national
demographics and health statistics.
Population parameters are based on the total EU population and on the population at risk
with the highest weight on national health expenditures. In the latter case, the share of
people suffering from diabetes, asthma, COPD, and heart failure represents the
population at risk.
Costs and benefits parameters are represented by estimates derived from the information
gathered through the literature review. The publications under review were scrutinised for
parameters indicating costs, benefits and net benefits under traditional and telemedicine
treatments. The parameters collected were aggregated through summary statistics and
applied on the population parameters and health survey information to provide a
descriptive economic analysis of costs and benefits from the societal and healthcare
provider perspective.

Market study on telemedicine
Final Report
118
In addition, the specific variables and parameters used in the decision model were
selected to fit the logic of the model. These variables and parameters are:
The population of the EEA (EU 28, Norway, Iceland and Lichtenstein), both the total
population and the share of individuals suffering from the above-mentioned four
chronic diseases.
The propensity to use remote technologies to consult a doctor gathered from the
Eurobarometer survey N° 460 (2017) on the attitudes towards the impact of
digitisation and automation on daily life, which comprises a study of the population
attitudes on digital health and care.
The average number of consultations observed for the population.
The average price of a consultation under both traditional and remote means, gathered
from the figures collected from the literature review.
The estimate of total minutes of consultation under both traditional and remote means,
gathered from the figures collected from the literature review.
The mortality rate under traditional and telemedicine treatment observed from the
literature review.
The total QALYs gained for traditional and telemedicine treatments observed from the
literature review.
Estimating cost-effectiveness of telemedicine with respect to traditional
approaches
The estimation of costs and benefits for each scenario described in the previous section
was carried out through the implementation of a decision model whose objective is to
simulate the patient journey from an initial consultation to a final treatment and
monitoring by means of traditional and remote (telemedicine) healthcare services.
The different possible stages of the patient journey decisions are visualised as a decision
tree comprising two initial branches, one for telemedicine/teleconsultation and a second
for traditional medical visit/treatment. These two branches are then split into three
possibilities of consultation frequencies: one, two, or several consultations.
In each of these branches, we have used as quantification parameters the population
likely to fall under in a given branch, the cost of consultation, and the time of
consultation. At the final stage of the decision model, we provide an outcome for the
patient journey in terms of success and failure and compute the total costs and the total
benefits of the journey associated with each path.
The split of the population moving throughout each branch of the model is the result of
the computation of population shares using different parameters drawn from the
literature. The model output comprises summary statistics determined by the population
likely to be in each final branch. This output comprises the following aggregates:
Total cost of the patient journey expressed in euros;
The total QALYs gained;
The total consultation time expressed in days;
The potential lives lost expressed in head counts and based on the mortality
parameter;
The total distance saved expressed in kilometres.
The figures below and overleaf provide an illustration of the economic decision model for
the case of the deployment for the full population and the case of deployment for the
chronically ill population; both under the baseline scenario (business as usual), and the

Market study on telemedicine
Final Report
119
alternative scenario (efforts to increase adoption by 5%). These figures also show how
the economic decision models are based on the information collected during the literature
review and other statistical sources.
Full population - Baseline
Full population – Alternative Scenario
Figure 49: Evaluation model on the total population, decision model’s telemedicine branch
Chronically ill population - Baseline
Chronically ill population – Alternative
Scenario
Figure 49: Evaluation model on the chronically ill population, decision model’s telemedicine branch
4.2.4. Summary of results
The present section presents a summary of the results drawn from the CEA analysis of
future deployment of telemedicine solutions and services across the EU. These results
include a comparison between the costs and benefits evaluated under the baseline
scenario (business as usual) and those evaluated under the alternative scenario (efforts
to increase the adoption rate by 5%). They are presented for each of the two models
under investigation: deployment focusing on the total population, and deployment
focusing on the chronically ill population.

Market study on telemedicine
Final Report
120
The case of a full deployment for the total population
After application of the decision model on the total population, the results on the cost-
effectiveness analysis on the baseline scenario (business as usual) indicate that
telemedicine healthcare services dominate traditional care today, at least under
the assumptions of the present model, which by simplicity, focuses on the recurrence of
consultation. Indeed, telemedicine is driving down the total costs of healthcare
services for society; today, the mix of telemedicine and traditional medical approaches
to patient management and monitoring is of 18% to 82% percent of the population
respectively. The total costs based on the full population are lower with the current
proportion distribution between telemedicine and traditional approaches (126.4 euros)
than the total costs if there were no telemedicine (143.3 euros). In this case, the general
costs per patient (standardised by the EU population including the EEA area) represent
8.8 euros per individual under the telemedicine and 117.5 euros under the traditional
approach.
Furthermore, other benefits include the amount of QALYs gained, which are still superior
under the use of traditional medical approaches, the time of consultation expressed in
days, which is higher for the traditional medicine approach, the mortality rate, and the
distance saved. According to these results, telemedicine is cost-effective with respect to
traditional medical services in terms of costs, logistics, and mortality.
The results from the evaluation of costs and benefits under the alternative scenario
(increase adoption by 5% rate) indicate that an increase of 5% in the utilisation rate of
telemedicine services and tool by the total European population (including the EEA area)
is cost-effective. Indeed, the share of the population going through telemedicine accounts
for lower patient journey total costs with respect to the share of the population going
through traditional medicine approaches. The total costs (standardised by the total EU
and EEA area population) associated with the 23% of the population using telemedicine
amounts to 11.31 euros per inhabitant, while the total costs associated to the 77% of the
population using traditional medical approaches amounts to 110.39 euros per inhabitant.
In total, the cost for society associated with an utilisation rate of 23% of telemedicine is
of 121.7 euros per inhabitant.
When compared with the baseline scenario, an EU intervention to spur investments and
efforts to increase the current adoption rate by 5% is preferred to the baseline in the light
of the difference between societal costs of 126.4 euros per inhabitant under the baseline
and 121.7 euros per inhabitant under the full deployment scenario.
In addition, in terms of benefits we observe the following evolutions with respect to the
baseline scenario:
An increase of 1.7% QALYs gained per inhabitant,
A decrease of 1.7% time spent in consultation,
A decrease of 3.6% in the mortality rate in case of failure, and
An increase of 27.8% in distance saved.
These results indicate that the democratisation, awareness raising and final uptake of
telemedicine services can provide considerable benefits for society by improving the way
healthcare service are organised and delivered. The human factor will always be
important in any patient-doctor interaction, but logistics benefits can considerably drive

Market study on telemedicine
Final Report
121
costs down enabling the medical personnel to focus on the most valuable tasks, hence
improving the quality of life of the population.
Table 4 : Cost-effectiveness results from the baseline scenario (standardized by the total population
– EEA area)
Full population -
Baseline
Tele-
medicine
Traditional
Medicine
ICER (for
total
benefits
only)
Total
scenario
costs/ben
efits
Total costs for
100%
traditional
medicine
Cost of patient journey
8.85 €
117.55 €
126,41 €
143,36 €
QALYS gained/individual
0.0323
0.1082
1,432.39 €
0,1406 €
0,1320 €
Days of consultation/
individual
0.0042
0.0277
4,620.54 €
0,0319 €
0,0338 €
Mortality%
0.0815%
1.0411%
11,327.98
€
0,0112 €
0,0127 €
Distance (Km)/individual
0.1408
0.0000
772.11 €
0,1408 €
0,0000 €
Table 5: Cost-effectiveness results from the partial deployment scenario (standardized by the total
population – EEA area)
Full population –
Increase adoption by
5%
Tele-
medicine
Traditional
Medicine
Total
scenario
costs/bene
fits
Evolution
with
respect to
baseline
Telemedicine
preference
Cost of patient journey
11.31 €
110.39 €
121,70 €
-3,7%
Yes
QALYS gained/individual
0.0413
0.1016
0,1430
1,7%
Yes
Days of
consultation/individual
0.0053
0.0260
0,0313
-1,7%
Yes
Mortality%
0.1042%
0.9776%
1,0818%
-3,64%
Yes
Distance (Km)/individual
0.1799
0.0000
0,1799
27,8%
Yes
The case of a partial deployment focusing on the chronically ill
population
Under the model focusing on the chronically ill population, the results from the analysis
on the baseline scenario also indicate differences between the total costs for society. The
total costs under the current situation (mix of 18% share of the population suing
telemedicine approaches vs. 82% using traditional approaches) are lower than the
total costs in a situation with 100% individuals using traditional approaches
(81.2 euros and 89.4 euros respectively), indicating a clear dominance of telemedicine
solutions over traditional approaches for diagnosis, treatment and monitoring. In this
case, the general costs per patient (standardised by the EU population including the EEA
area) represent 7.8 euros per individual using telemedicine tools and services and 73.3
euros using traditional approaches for diagnosis, treatment and monitoring.
Furthermore, in the case of analysis under the alternative scenario (increase adoption by
5% rate), and according to the proportions on the frequency of consultations observed
from national statistics data, we compute the proportions associated with one, two or
several visits to a doctor for individuals suffering from the specific chronic diseases. The
economic evaluation under the alternative scenario assumes that 52% of the chronically
ill population visits a doctor at least once during a year, 26% visits a doctor at least twice,
and 22% visits a doctor three or more times.

Market study on telemedicine
Final Report
122
The results from the evaluation of costs and benefits under the alternative scenario
indicate that telemedicine dominates traditional medical approaches throughout the
patient management and follow up process. Indeed, the overall cost for society
(standardised by the total EEA population) for the patient journey is of 10 euros per
inhabitant while under traditional medical journeys it is of 68.9 euros per inhabitant. In
addition, the time spent on consultations is also lower in the case of telemedicine (0.003
days per inhabitant vs. 0.014 days per inhabitant). In addition, other benefits are also
superior in the case of telemedicine approaches to patient management and monitoring.
These results indicate that investments and awareness raising efforts made to increase
the uptake of telemedicine is cost-effective under a scenario focusing on the chronically ill
population.
With respect to the baseline scenario, a focus on chronic diseases and the population
suffering from them indicates that actively investing in the uptake of telemedicine, at
least by 5%, is cost-effective as the evolution of total costs savings and benefits
(standardised per inhabitant) improve the situation. Indeed, the total costs of a mix of
23% telemedicine and 77% traditional medical approaches, with respect to the baseline
scenario (18% to 82%) are lower by 2.8%, while in terms of benefits, QALYs gained
increase by 1.7%, the time spent on consultation decreases by 1.7%, the mortality rate
decreased by 3.6% and total distance saved by 27%.
Table 6 : Cost-effectiveness results from the baseline scenario (standardized by the total population
– EEA area)
4.3. General conclusions
The analysis of costs and benefits from the wide deployment of telemedicine indicate that
no matter the path chosen by decision makers, the main benefits and savings will rather
be in the hands of society, mostly citizens and municipalities through logistics savings and
productivity gains. Indeed, even if the time of a consultation, as adopted by the decision
model, is in average 14 minutes, it usually costs the patient about a half or a third of the
day in productivity, hence increasing the burden on society through the reduction of the
economic activity. However, with telemedicine solution, the effective productivity losses
are associated with less than 30 minutes away from economic activities.
This distribution of benefits seems uneven, making it difficult to explain why healthcare
systems should heavily invest in technologies that will only provide benefits for the
patient and not the institutions or hospitals themselves. However, as demonstrated
above, national health systems can benefit from productivity gains from equal levels of
expenditure at a higher quality of service and at an increased time spent by human
resources focus on value added care.
The preference for a wide deployment of telemedicine implies the need to find new ways
to deliver high quality care services to optimize the organization of health systems to
reduce the scope of non-value-added activities through the proper use of telemedicine
technologies. The organizational change has the potential to optimise consultations from
the perspective of a personalised care approach taking into account the real needs of a
patient.
In general, the outlook of a wide telemedicine deployment seems positive and promising
because there is an increasing awareness of the need; today the EU is in the very early

Market study on telemedicine
Final Report
123
days of adoption, and just the fact that there is an absence of a harmonized
reimbursement system indicates that it is far away from full potential. However, individual
Member States are moving forward with the UK promoting teleconsultation as a way to
save costs for the NHS while in France teleconsultation is going to be reimbursed from
September at the same amount as for a physical visit to the doctor (€25 for a general
practitioner and €30 for a specialist doctor)
100
.
Yet, it is necessary to involve the companies driving innovation in the actual design of
processes, such as regulatory processes with CE Mark. This is necessary in order to
ensure that innovation is being promoted. In order to spur efforts for the uptake of
telemedicine, it is necessary to better assess what is useful for society in terms of cost-
effectiveness based on scientific evidence as well as people’s perceptions. Indeed, there
are occasions where tools from big companies that are not scalable and therefore only
benefit a few at high costs find support for deployment, while scalable or more useful
tools from innovative SMEs do not. Today, there are non-for-profit organisations trying to
make the voice of innovative start-ups heard on the policy making field, to proactively
involve them in the process of EU harmonization.
What is also important is raising awareness about the potential benefits of the adoption of
telemedicine, to the public and their representatives (elected politicians), policy makers
and those working in health related professionals.
Conclusions from the analysis of primary data
From our discussion with key experts in the telemedicine field, we concluded that some
challenges offer opportunities for assisting the wider deployment of telemedicine. One
notable example is the need to make a bigger effort to encourage heterogenic groups of
professions (medical doctors, nurses, ICT professionals in health) to develop a common
language so that can integrate better, develop a holistic understanding of the needs of a
patient and help in the design and deployment of telemedicine solutions.
One way to achieve the smooth collaboration of different groups is for them to meet in
workshops so that they can communicate, understand each other and generative
innovative ideas through brainstorming. One such idea would be regulators, nurses and
medical doctors to develop a data bank (in which patients can find accurate medical
information), a collaborative platform for medical professionals to share knowledge, etc.
The EU and individual Member States could financially support and encourage the sincere
dialog between different stakeholders. Such dialogue would allow stakeholders to
understand each other, especially now that the digital disruption and the increased
complexity of systems makes it difficult for individuals to follow and adopt.
Another example of action that would increase the use of telemedicine is to simplify the
tools at the development stage so that all medical and paramedical personnel can actually
use them within the frame of their known procedures. This also need a significant degree
of communication and coordination.
Finally, as new technologies allow more efficient treatment, it is the responsibility of all
stakeholders to test, adopt and put the new approached in production. One example is
that it is now possible to make a heart surgery with alternatives to an open operation,
100
https://www.connexionfrance.com/French-news/French-healthcare-to-cover-remote-consultations

Market study on telemedicine
Final Report
124
using more efficient techniques enabled by the recent technology. This has reduced the
post-surgery stay length for the patient from 8 to 3 days demonstrating a clear benefit
for both the patient (recovery speed) and the health care system (cost reduction, treat
more patients).

Market study on telemedicine
Final Report
125
5. CONCLUSION
The following main findings and considerations can be derived from the analysis
conducted in chapters 1, 2, 3, and 4. These represent the key messages for decision-
makers to keep in mind when considering further policy developments or initiatives at EU
level to support telemedicine uptake, if desirable.
5.1. Main findings
Finding
1
Solutions: The telemedicine solutions landscape is concentrated in
a number of MS, and primarily targets the telemonitoring and
prevention space in primary care, and in relation to main chronic
diseases. A great number of solutions target well-being and self-
care, especially mobile health applications.
Most telemedicine solutions available on the market are deployed at national or regional
level, while few are in use in multiple MS or have international penetration beyond the
EU. This is due to the significant differences in national regulations and social security
schemes (see “Barriers” below). While these remain a national competence,
interoperability between solutions – which is another challenge to cross-border use –
can be addressed by policy-makers at EU level and should remain a priority in
the coming years, to stimulate the market. Still, the majority of solutions have been
in use for over five years, which testifies to stable demand and future potential.
Software and medical devices are the predominant solution types, but typically, solutions
comprise several components: hardware, a supporting platform, application, database
and/or services. Considering a ‘solution’ as an entire ecosystem and the related data flow
end-to-end are also key in effective regulation and market facilitation. Therefore, a new
‘solution’ definition may be required to reflect this complexity. At the same time,
different companies offer the individual solution components, so fragmentation
between legal frameworks regulating different components must be eliminated.
The proliferation of well-being solutions, i.e. those that are not related to a (prior)
medical condition/disease, suggest that there are more possibilities in this segment at
present, while wider uptake of telemedicine for monitoring, prevention and treatment of
medical conditions is to be expected in the longer term. Specific challenges and policy
recommendations related to these types of solution merit a separate analysis, as
they were outside the scope of the present study.
Finding
2
Standards and guidelines: The applicable standards and
guidelines mostly address technical requirements.

Market study on telemedicine
Final Report
126
It is not surprising that most standards and guidelines are set by international bodies and
are of a technical nature, given that technology is at the core of telemedicine practice.
However, at times these are not specific enough; in these cases, Member States set their
own national standards, especially to provide precise requirements for telemedicine
solutions related to given medical specialties. It is important to tackle this going forward,
especially as it directly links to the need for interoperability. In this regard, the role of
EU policy makers would also be to:
Better understand current limitations and needs,
Offer more detailed specifications,
Work with the entire range of stakeholders (SDOs, market players, healthcare
providers, etc.),
Raise awareness and support capacity-building related to the use of standards and
guidelines,
Link legislative effort with necessary supporting measures related to standards and
guidelines.
Beyond standards definition, testing, classification and certification processes are
also essential to prepare the deployment of telemedicine services on a large
scale.
As regards other types of guideline/rule, there seems to be good coverage of all relevant
domains at present: data protection, organisational, human resources, ethical and EHR.
Finding
3
The market: The market potential of telemedicine is strong. It is
expected to grow at a compound annual growth rate of 14% in the
coming years. The well-being market enabled by digital
technologies (mobile applications, devices) is rapidly growing as
well.
The uptake of information technologies in Europe, which has enabled the remote
transmission of information at ease, speed and at marginal cost, is the main accelerator
telemedicine, which is currently experiencing rapid growth. This is facilitated by two key
preconditions: access to the technology or infrastructure, and favourable financial
conditions for telemedicine programmes.
Indeed, we observed that demand outpaces supply, but this should be read with care, as
there are many telemedicine initiatives but adoption is at early stages, since hospitals and
clinics are in demand of these solutions, but do no currently have the appropriate means
to pay for the technology by leveraging on well-designed reimbursement systems. Here,
there is only a limited role for EU decision-makers in harmonising approaches and
enabling reimbursement schemes at EU level to facilitate adoption. However, there is a
role for the European Commission in identifying good practices in MS related to
funding schemes for telemedicine and in supporting their adoption by other
interested countries.
A wide range of market players is active, including: telecommunication companies, ICT
tools and electronics manufacturers, device manufacturers, pharmaceutical industry
companies, and start-ups. They are concentrated in countries with relatively high

Market study on telemedicine
Final Report
127
healthcare expenditure per inhabitant, and we note a trend towards partnerships between
healthcare and technology players. This and other business models should be
studied further by policy-makers to enable better regulation.
Finally, it emerged from our research that the US and Canada have outperformed the EU,
whilst Japan has the lowest volume of users of telemedicine. Factors that contributed
to success in North America and a lag in Asia are relevant, and exchange of
experience at policy-maker level should take place.
Finding
4
Barriers: Difficulties relating to access to telemedicine in Europe
exist in all countries, with: the lack of acceptance of telemedicine
solutions by stakeholders; the unfavourable regulatory framework;
the insufficient funding; and the inadequate IT infrastructure being
the most prevalent ones.
Decision-makers should be attentive towards barriers preventing the wide deployment
and adoption of telemedicine, and pursue actions to overcome these (an exhaustive list of
proposed countermeasures is include in Chapter 3 and will thus not be repeated here). In
particular, conservatism or resistance to adopting new medical processes, integration
between technology and medical practitioner’s procedures, and (data protection)
regulations are delaying the generation of the base of evidence necessary to convince all
actors, and EU level policy-makers should prioritise these actions in the upcoming period.
In addition, attention should also be given to regulatory approval for solutions, to
streamline and make it less costly.
If not addressed, current barriers will delay the deployment and adoption of telemedicine
solutions in Europe by years, and as a consequence, the EU also risks seeing market
players going to the US or other more favourable economies.
Uptake of telemedicine solutions across national health systems will also only be
successful if key institutions in the medical community, such as recognised clinics and
hospitals, establish new partnerships. These institutions will only be incentivised to do so
if national decision-makers allow health systems to properly pay the utilisation of the
technology, meaning developing reimbursement schemes for telemedicine utilisation.
Further to this point, it is important to highlight that today, only direct consumer models
have some degree of success, only because institutional players cannot pay for or are not
always reimbursed for telemedicine tools and services.
Finding
5
Cost factors: Telemedicine is generally perceived and judged to be
cost-effective, as evidenced by trials documented in academic
literature.
Our systematic review of the reported cost-effectiveness of telemedicine was carried out
by means of a structured inventory of the existing published data and statistical
comparisons.

Market study on telemedicine
Final Report
128
In essence, we confirmed that telemedicine is reported cost-effective in 73.3% of the
cases covered by the literature. Neutral effects were discussed in 21.3% of the selected
references, mainly in systematic reviews. Negative effects account for 5.6% of the
selected studies.
An additional outcome of this review was the identification of cost factors or cost
parameters, which have strong impact on the cost-effectiveness of telemedicine solutions.
These include: distance between patient and nearest healthcare professional; time
required per consultation; cost of a doctor visit; QALY; mortality rate. The values of these
cost parameters prove to directly affect projected cost-effectiveness of telemedicine
solutions. Indeed, we used the cost factors identified as a basis in performing a cost-
effectiveness analysis of the deployment of telemedicine on a wide scale.
The literature suggests that telemonitoring solutions are proven or indicated as being the
most cost-effective in relation to a broad range of diseases, with an emphasis on chronic
medical conditions.
While the review shows robust results, they are linked to individual solution trials, and not
to wide-scale solution deployment. Hence, broad conclusions regarding cost-effectiveness
of telemedicine based on this study of literature should be made with caution.
Finding
6
Large-scale deployment: Further adoption of telemedicine is cost-
effective, though benefits from wider uptake will be tangible for
patients and society at large through logistics savings and
productivity gains, and less so for healthcare providers or social
schemes. More scientific evidence is needed from larger scale trials
and telemedicine programmes to conclude on this definitively.
In general, the trend of telemedicine adoption will continue to increase and is likely to
generate considerable savings and benefits for society, though adoption is at an early
stage and it may take some before wider uptake can be measured.
Under two scenarios: one, of telemedicine adoption at a rate of 18%, and another –
where we simulated telemedicine would be preferred by 23% of the population, we
compared the costs and benefits of telemedicine to the traditional face-to-face patient
journey. To further differentiate possible alternatives, the scenarios looked at the cost
efficiency when telemedicine would be used by the population of people suffering from
chronic diseases, and by the total population of potential patients. We also examined
various scenarios for up to a 48% share of telemedicine. We were able to observe clearly
that the higher the share of telemedicine – the more cost-effective wide-scale deployment
becomes. An increasing share of telemedicine decreases the total cost of the patient
journey, total consultation time, distance travelled and mortality rates, while increasing
QALYs gained.
Further adoption of telemedicine increases benefits: it reduces costs (consultation costs,
travel costs, time spend) and increases patient survival and life quality. However, policy-
makers need to invest in obtaining more scientific evidence for its efficiency and large
scale experiments to assess the impact of a wider deployment. Raising awareness
(patients, doctors), stimulating integration between stakeholders and reimbursement are
keys to speeding up success.

Market study on telemedicine
Final Report
129
5.2. Additional considerations
The opportunities for the deployment and adoption of telemedicine across the EU are
triggered by a changing demography of both individuals and medical professions; these
changes call for a search for optimization of the healthcare service provision driven by the
perspectives of self-management and prevention. In consequence, the EU needs to
have an ambitious target with respect to implementation and deployment.
Policy designs need to take into account the specificities of each country and region,
providing not only the idea and vision and strategy, but also narrowing them down to
actual projects that can reflect tangible benefits for the public. When assessing the
uptake of telemedicine across different countries, with different cultures and technology
maturity, projects need to be tailored to the specificities of the country and the interests
of the local population. Although common interests across EU populations can focus on
two groups of people:
The elderly and the chronically ill population which will drive telemedicine
consumption, and
The young or healthy population that cans benefit from prevention and management to
stay healthy and economically active.
The benefits must be communicated at local level since it is potential patients who need
to be convinced about the utility of telemedicine. Raising awareness among the
stakeholders of the value chain of telemedicine and the public is key and the European
Commission can play an important role in supporting this exercise.
Indeed, citizens need to be better informed about the novelty of the technology and how
it will impact them; if possible, changes to the national healthcare systems need to
integrate a degree of co-development with different actors of the process, decision
makers, market players and institutional and end users.
Today there is no clear set-up on the adoption model the EU wants; whether it is based
on performance improvement or better quality of service, decision-makers will still need
to make it clear to generate enough confidence for the adoption process to take place.
The fundaments of interoperability, secured data transmission, storage, handling
and accessing permissions for health data are not clearly defined either; even if new
regulations in place have set clearer rules about private data, these are still not tailored
for patient data.
Telemedicine has been discussed for nearly 40 years, but even if today the technology
and social conditions for its uptake are met, there is still a lack of evidence to support
it. When taking national decisions affecting directly the health of the population, decisions
need to be taken on the basis of scientific facts. At present, we lack this base of evidence
to prove the effectiveness of telemedicine.

Market study on telemedicine
Final Report
130
Another key consideration is the importance of the human factor. Face-to-face
interactions are necessary and relevant in clinical caregiving, therefore any discussion
about the widespread deployment of telemedicine should consider this.
The dynamics of the medical profession, including nursing, is pressing governments to
accelerate adoption, but barriers on the use by the profession are still standing, whereas
all medical professions will need to have the competence and knowledge to do
telemedicine and telenursing. As an example, from the professional perspective in several
countries, nurses are not allowed to perform certain medical procedures (e.g. give advice
to patients, prescribe); this represents a barrier closely related to the professional
culture, procedures, and data handling permissions. This represents a weakness for the
national health system. Convergence of the medical and paramedical professions in
terms of training can trigger the change of culture to solve this weakness. In addition,
better communication between technical developers and the medical
professions would be necessary.
In essence, telemedicine promises a huge potential for patients and society as a whole. It
will help improve the quality of diagnosis, treatment and quality of life throughout the
patient management process.

HOW TO OBTAIN EU PUBLICATIONS
Free publications:
• one copy:
via EU Bookshop (http://bookshop.europa.eu);
• more than one copy or posters/maps:
from the European Union’s representations (http://ec.europa.eu/represent_en.htm);
from the delegations in non-EU countries (http://eeas.europa.eu/delegations/index_en.htm);
by contacting the Europe Direct service (http://europa.eu/europedirect/index_en.htm) or
calling 00 800 6 7 8 9 10 11 (freephone number from anywhere in the EU) (*).
(*) The information given is free, as are most calls (though some operators, phone boxes or hotels may charge you).
Priced publications:
• via EU Bookshop (http://bookshop.europa.eu).
