
International Program UAM-Boston University
Laboratory Manual
Organic Chemistry I
2013-2014
Departamento de Química Orgánica
Ernesto Brunet Romero
Ana María Martín Castro
Ramón Gómez Arrayás

1
Laboratory Manual
Table of Contents ............................................................................... 1
Introduction ............................................................................... 2
Prelab preparation ............................................................................... 2
Notebook ............................................................................. 3
Safety .............................................................................. 3
Laboratory Practices and Safety Rules ............................................................. 4
Accidents and injuries ........................................................................... 5
Fires ............................................................................. 5
Chemical Wastes ............................................................................. 6
Cleaning Responsibilities ............................................................................. 6
Lab cleanliness ............................................................................. 6
Laboratory Equipment ............................................................................. 7
Proper use of glassware ............................................................................. 8
Some techniques in lab experiments
Heating, cooling and stirring ............................................................................. 9
Measurements ............................................................................. 10
Extraction ............................................................................. 10
TLC chromatography ............................................................................. 13
Recrystallization ............................................................................. 18
Common organic solvents ............................................................................. 23
Experiment 1: Using Extraction and NMR to isolate and identify the analgesic active
principles of a sample ............................................................................. 24
Experiment 2: Preparation of an azoic dye: Para-red ........................................... 35
Annex to Experiment 2: Acetanilide nitration: a 1H-NMR exercise ……. 43
Experiment 3: Synthesis of a flavoring principle: Isopentyl Acetate ................... 47

2
Introduction
In this laboratory course you should be prepared to apply and understand what you have learned in
General Chemistry and the Organic lecture course to real situations. You should never expect to walk into
lab and see something illustrated from class. That is not the way science works. Laboratory is for discovery!
You will expand your theoretical understanding (microscopic) to the macroscopic scale. Whether or not
organic chemistry is your idea of fun, there is no reason for you not to enjoy your organic laboratory
experience.
Lecture and lab are, of course, related. Chemistry is an experimental science. Everything you learn
in lecture was originally discovered in a laboratory. When asked what ethanol is, many chemists, both
students and teachers, are satisfied with the answer “CH
3
CH
2
OH”. This answer is wrong, or at best,
incomplete. To answer the question “What is ethanol?” you have to know that it is a clear liquid, flammable
and a powerful intoxicant. Ethanol is an actual substance and “CH
3
CH
2
OH” is simply one convenient model
we use for describing it. Never forget the substance behind the formula! Is it a solid, liquid, or gas? Is it pure
or a mixture? Is it life-sustaining or lethal?
There are two overriding principles that guide this organic laboratory curriculum: (1) Laboratory
should present the opportunity for you to learn how to extract knowledge from an experimental result,
because that is the heart of science. (2) Laboratory should engage you into learning about chemistry.
Organic reactions rarely lead to a single clean product in stoichiometric yield, so the ability to find
out what the product of a reaction is depends on your ability to purify the material. Much of the new learning
is centered on methods of purification and analysis. All of this requires learning new laboratory techniques.
Upon completion of this laboratory course, you should have an understanding of a selection of tools and
techniques used by organic chemists. Aside from good laboratory technique you should strive to learn how
to take data carefully, record relevant observations, and use your time effectively. You will be asked to
assess the efficiency of your experimental method and to plan the preparation, isolation, and purification of
organic substances.
The development of your scientific writing and record keeping skills is an important aspect of this
course. It is anticipated you will improve your skills in communicating analytical results in a clear and
concise manner.
Finally, you should understand the need for safe laboratory practices involving chemicals and their
conditions for use.
Prelab preparation
Advance preparation for lab is one key to success in the organic lab. Unprepared students waste
time [their own and the teaching assistant’s (TA)] and can be a hazard in the laboratory. Before the start of
lab,
- You should read appropriate sections in the lab manual and online sources.
- You should be certain of the purpose of the experiment.
- Information regarding references to any literature used should be prepared in the laboratory
notebook.

3
- A plan of action should be outlined taking into consideration reactions, hazards and important
physical data.
Notebook
Use a bound notebook with pages pre-numbered by the manufacturer. Leave a few blank pages at
the beginning of the book to build a table of contents when you finish the lab.
What You Should Write in Lab
You should record the following while an experiment is progress: procedure, data, observations,
and those conclusions on which an immediate procedure decision is based. All notebook information
should be as complete as possible. If necessary, you or another student should be able to refer to your
notebook and repeat the experiment exactly, comparing observations to those on record. The body of the
notebook must be written as simultaneously as possible with your performance of the lab work.
What You Should Write After Lab
Occasionally you will analyze data, find information in reference books, share data with another
student, interpret spectra, and calculate stoichiometry, etc. after lab is over. This information, along with the
answers to post-lab questions, should be kept in your notebook.
The Mechanics of Keeping a Lab Record
For the part of your notebook to be kept in lab, all entries should be made directly into it and not
copied from another source. NEVER record elsewhere information that belongs in the notebook neither
on handy scraps of paper, margins of the lab manual, filter paper, or paper towels. NEVER temporarily
memorize information for later transcription into your notebook. Do not be overly concerned with the
cosmetic appearance of your notebook. While certain amount of neatness and organization is necessary,
legibility and comprehensibility are the essential qualities.
Be careful not to render conclusions in your notebook (e.g., “the boiling point is…”) without the
supporting data or observations, i.e., “Bubbling slowed as the temperature dropped to 120-115 °C. Bubbling
stopped and liquid entered the tube at 114.5 °C. Tube full of liquid at 113 °C.”
Safety
Safety in the laboratory is an extremely important element in the chemistry program at this
University. Failure to follow safe practices can cause laboratory accidents that may result in personal injury
or, at the least, loss of time, damage to clothing and other property. By following suitable precautions, you
can anticipate and prevent situations that could lead to accidents. You must become thoroughly familiar
with the information in the following sections, as well as the specific information provided for each
experiment. You must also sign the Laboratory Safety Rules and Practices Contract that you will receive in
the first scheduled laboratory meeting.
Eye protection (safety goggles) is mandatory for all occupants of the laboratory when anyone is
performing lab work.
4
Dispose of all chemicals properly. Liquid and solid waste should be disposed of in the designated
WASTE bins and/or tanks. The lab has a special waste bin or bottle for each special type of waste. Please
be aware of any waste instructions in the pre-lab lectures. If you are unsure, please do ask your Teaching
Assistant.
Should a chemical spill occur, please clean it up at once with the appropriate technique. We all get
mad when we hear about some chemical company that creates a spill and is slow at solving it. This is true
at the undergraduate laboratory as well. Spills occur frequently at the balance area. You ought to make
sure that you do clean up your chemical spill. If you do not know how to clean up a particular spill, notify
your lab TA.
Fire is of maximum concern in the organic laboratory. While heat guns are available in the lab, use
them only in the hood after ensuring the area is clear of flammable liquids. Be aware of your surroundings
at all times. Look around your lab room and make sure that you are aware of the location of the fire
extinguishers, showers, and eye washers.
Laboratory Practices and Safety Rules
A. Personal Protection
1. You are only allowed to work in the laboratory if, and only if, the teaching assistant is present.
2. You must work only on authorized experiments.
3. You must wear proper eye protection in the laboratory whenever any laboratory work is in
progress.
4. You must wear shoes that do not have open spaces; sandals, flip-flops or any peep toe shoes
are not acceptable.
5. You may not eat, drink or smoke in the laboratory. You must not even bring food or drink into the
laboratory.
6. You must confine long hair and neckties. Loose jewelry may also be a hazard.
7. You must not engage in acts of carelessness while in the laboratory.
8. You must work carefully with a full awareness of what you are doing in order to avoid ruining
equipment or spilling chemicals.
B. Proper Laboratory Practices
1. Carefully read TWICE the label on a bottle before using its contents.
2. Take only the quantity of reagent needed. NEVER return an unused reagent to its container.
3. Mix reagents only when specifically directed to do so.
4. NEVER place chemicals directly on the balance pan. Weigh reagents using a beaker, flask or
weighing paper.
5. If instructed to observe the odor of a chemical, do so by fanning air with your hand over the
container toward your nose. DO NOT directly smell any substance.
5
6. The fume hood is for your personal protection. You must leave the hood at the indicated working
level for your protection and the protection of others. Do not lock the hood in the full-open
position. The air-flow velocity is insufficient when the hood sash is in the fully-raised position.
7. NEVER taste reagents.
8. Avoid handling chemicals directly with your hands. Protect your hands with gloves. If contact
occurs, immediately flush the area with plenty of water.
9. Use a bulb or a pipetting device to draw liquids into a pipette. NEVER do pipetting by sucking
with your mouth.
10. When diluting strong acids or strong bases, the acid or base should be added to the water, not
the other way around.
11. Try to avoid using heat guns but before turning it on, make ALWAYS sure no flammable liquids
or vapors are close in the area.
12. Heat test tubes at the surface of the liquid. Agitate the tube. Be sure to slant its open end away
from yourself and other people.
13. Stay clear of an open vessel in which a process is occurring that could produce spattering.
14. Keep reagents and equipment away from the edge of the lab bench.
15. Do not use cracked glassware, as it may break when even slightly stressed.
Accidents and Injuries
You must report all accidents and injuries to the TA as soon as possible. Band-aids and first aid kits
with some simple medical supplies and latex gloves are located in the laboratory. Wear gloves when
helping with an open wound. In the event of an injury, some basic first aid procedures should be
immediately carried out as follows:
- Skin Burns or ocular lesions: There are more than 25,000 chemicals likely to cause skin or
ocular lesions and burns (after a single or repeated contact), such as acids, bases, oxidizers,
reducing agents, and solvents. The affected tissues must be rinsed as quickly as possible with
DIPHOTERINE
®
solution (BE AWARE of its location in the lab) as a first-aid treatment in
emergency situations at the workplace (or at the scene of the accident). The sooner the first-aid
is applied, the lower the probability of any serious after-effects. It stops the development of
chemical burns and allows a rapid return to a physiological state.
- Hair or Clothing Fires: Use quickly the safety shower to extinguish flames.
Fires
- If the fire is contained in a beaker, try to smother it with a fire blanket placed over the beaker.
For a larger fire, discharge the fire extinguisher at the base of the flame.
- In the event of a large or uncontrollable fire, TA’s must direct students to immediately evacuate
the room, according to the following evacuation procedure:
a) Direct students to leave the building (BE AWARE where the emergency exits are
6
located)
b) Shut down all equipment in the laboratory, if possible, and close all doors
c) Activate the fire alarm in the hallway
a) Report the fire to the authorities, or call emergency (112)
Chemical Wastes
Special instructions for waste disposal are given at the end of each laboratory procedure. Properly
dispose of all wastes: the trash can, sink, glass disposal box, solid waste disposal box, or hazardous liquid
waste bottle will be properly designated. Please, BE AWARE of their location. Never pour organic solvents
or toxic wastes, such as solutions containing chromium, mercury or lead, into the sink.
Cleaning Responsibilities
1. You are responsible for cleaning any equipment used in the experiment, cleaning your
immediate work area, and returning equipment to the proper places.
2. Additional responsibilities for cleaning designated areas of the laboratory will be assigned by the
TA.
3. Clean all glassware before storing it. Soap solution and squeeze bottles of acetone for cleaning
are provided at the large sinks.
4. Neutralize acid, basic or neutral (organic) spills with the apprpriate solid absorption agent before
cleaning the area. For large chemical spills on the bench or floor, immediately alert your
neighbors and the TA. Clean the spill as directed.
5. Dustpans, brooms and brushes are available in the lab for sweeping broken glass from the
benches and floor. Place broken glass in the special cardboard containers provided (glass
disposal boxes).
6. Remove any paper, broken glass or any other debris from the sinks.
If you behave in an unsafe manner in the laboratory you will be elegible for immediate expulsion
from the laboratory. Unsafe behavior includes, but is not limited to, failure to wear proper goggles and
proper lab attire including proper shoes. If you are expelled in this manner you will not receive credit for the
experiment and will not be allowed to make up the experiment.
Lab Cleanliness
Clean-up begins 15 minutes before the scheduled end of the period. You are responsible for
cleaning up your personal work area. This includes returning all equipment and supplies (hot plates, ring
stands, clamps, etc.) to the proper place, correctly disposing of any waste, and cleaning the bench-top.
Failure to do this will result in loss of technique points.

7
Laboratory equipment
Short path distillation
head
Vigreux
distillation
Reflux
condenser
Distillation
receiver
Graduated
cylinder
Three-necked round-
bottom flask
Erlenmeyer
Flask
Prolonged
clamp
Solid addition
funnel
Separatory
funnel
Büchner funnel Kitasato flask Addition funnel
Liebig
condenser
Volumetric
flask

8
Cold finger Dessicator Beaker
Fritted glass
funnel
Distillation
termometer
Clamp holder
Reducing joint
adapter
Chromatography
column
TLC developing
tank
Round bottom
flask
Ring clamp
Rings/clamps
Stand
Hot plate stirrer Keck clips Stir bars
Proper use of glassware
Most of the experiments in this manual are described on microscale or miniscale. This generally
means working with between 50 mg and 2 g of material. For comparison purposes, a regular aspirin tablet
contains 325 mg of acetylsalicylic acid. Working with small amounts of materials highlights the importance
of working with clean glassware. If you prepare 50 mg of a product, which then picks up 10 mg of foreign
material from dirty glassware, the product is now significantly contaminated.
Transferring small amounts of material from one container to another requires care. You can avoid
unnecessary transfers with careful planning. When transferring solids between containers, losses are
unavoidable. Do the best job you can to scrape material out of the original container. Quantities of liquids

9
less than 5 or 10 mL should be transferred using a Pasteur pipette. Always hold a pipette right side-up.
Never ever invert a pipette. This contaminates the rubber bulb and the sample you are holding. Do not
attempt to pour small amounts of liquids. If the liquid in question is a solution of your product in an organic
solvent (for example, 100 mg of benzophenone dissolved in 2 mL of hexane), you can make a very efficient
transfer by pipetting the solution to a new container, then rinsing the original container with a bit of the
original solvent (hexane). Combine this wash hexane with the material first transferred.
Most scientific glassware can be viewed simply as a container with a specialized purpose. When
choosing glassware, keep in mind what will happen later. Does the material need to be heated, cooled,
refluxed? Will you be adding more material? Choose an appropriate-sized container. It makes no sense to
store 5 mL of liquid in a 250 mL beaker.
Cleaning glassware
Always wash glassware before the end of lab. That way, when you return to lab later, it will be
clean, dry, and ready to use. Usually, soap, water and a little elbow grease are all that is necessary. For
highly water-insoluble materials, it may be necessary to rinse the item with a bit of wash acetone, located in
squeeze bottles by the sink. While working in lab, if you find it necessary to wash and re-use a piece of
glassware, determine if the piece must be dry before use. Many students have used a considerable amount
of lab time carefully drying a piece of glassware only to then use it to hold water.
Heating, Cooling and Stirring
Efficient cooling is performed in an ice bath, which really means an ice-water bath. If you are
cooling a small container that can tip over, clamp the container in place. If it can tip over, it will tip over.
Please heed this warning.
Heating is performed on a hot plate. A heat gun is never used to heat an organic solvent. Flat-
bottomed containers (beakers, and Erlenmeyer flasks) can be heated directly on the surface of the hot
plate. Round-bottomed items such as test tubes and flasks are best heated in the hole of an aluminium
block placed on the hot plate. The hot plates surfaces respond slowly to changes made in dial setting. This
surface will warm and cool very slowly. The most effective way to stop heating a container is to raise it
above the surface of the plate or remove the container entirely. Be careful: the high setting on a hot plate is
often extremely hot. Do not pick up an aluminum block after it has been resting on the top of a hot plate.
You will receive a nasty burn. At the end of lab, leave the aluminum block in the hood with the heater, not in
your drawer.
The hot plates in lab are also equipped with a magnetic stirrer. Below the center surface of the hot
plate is a strong magnet mounted on an electric motor. Solutions on the heater-stirrer can be mixed by
adding a Teflon-coated magnetic stir bar or spin vane.
Small amounts of materials in a large test tube can be effectively mixed by rapid agitation in small
up and down strokes.

10
Bumping
Organic liquids have a tendency to super-heat when being heated in a glass container. The super-
heated liquid will then boil violently in a sudden fashion: a condition known as “bumping.” Bumping will
usually spray the hot liquid around the laboratory and on surrounding people. Bumping is easy to avoid. To
avoid bumping one of the following solutions can be applied:
1. Add one or two boiling stones to any liquid you are boiling. [Boiling stones, also called boiling
chips or Boilezers® are small chips of an inert porous material such as porcelain or carbon.
They act as a nucleation source for boiling to occur].
2. Magnetically stir the liquid.
3. In some cases you can constantly attend the boiling of a small amount of liquid in a large
container, where constantly swirling of the solution is possible.
Measurements
It is important to understand whether a measurement needs to be accurate or whether an
approximation will suffice. Reagents affecting the stoichiometry of a reaction are generally made accurately.
Solvent, solution, and reagent quantities used in gross excess can often be approximated (e.g., organic
solvents or aqueous solutions used for extractions).
Precise measurements are best made on a balance. Small amounts of liquids are either weighed
or measured by volume with a syringe. A graduated cylinder does not deliver sufficient accuracy for small
volumes of an organic liquid (less than a few milliliters). Hamilton's precision syringe, which you may have
in the lab, are designed to deliver highly accurate and precise volumes of liquids, but are quite expensive
and indicated for special cases. Electronic balances are a great convenience, but are also expensive and
fragile. To maintain calibration, never move the balance. Keep the balance area clean, removing any spills
immediately. Before use, depress the “zero” or “tare” button. Place an item to be weighed on the pan, and
record the mass in your laboratory notebook. Because you cannot place chemicals directly on the balance
pan, you will weigh reagents on a weighing paper or directly into the container in which you plan to use the
chemical. In this case, the mass of the empty container must be subtracted from the gross weight of
container and contents. This process is known as a tare. The balance can provide this feature
electronically. Place the empty container on the balance, and again, press the “tare” or “zero” button. The
weight of the container is automatically subtracted. Do not use this feature, however, if you need to re-
weigh the container and contents later in the experiment.
The graduations on the sides of beakers and test tubes provide a nice method for the estimation of
volumes.
Extraction
When something is extracted, it is pulled away from something else. For example, a dentist extracts
a tooth by pulling it out of your mouth. In chemistry, extraction is the physical process by which a compound
(or a mixture of compounds) is transferred from one phase into another. When you make tea or coffee, an

11
extraction takes place: the water-soluble components in the tea leaves or the coffee grounds are
transferred from a solid phase into a liquid phase (the boiling water). This is an example of a solid-liquid
extraction. It is also possible to partition the components of a mixture between two immiscible liquids (i.e.,
liquids that will not dissolve in each other and form two distinct phases when combined). This process is
called a liquid-liquid extraction.
There are two general types of liquid-liquid extractions:
- An organic solvent extraction in which an organic solvent with a high affinity for the desired
compound is used to extract the compound from another solution.
- An acid-base extraction, in which an organic acid or base is extracted from an organic solvent
by using an aqueous solution of an inorganic base or acid, respectively. A neutralization occurs
which converts the compound into an ionic, water-soluble salt, causing it to transfer from the
organic phase to the aqueous phase.
The choice of apparatus for an extraction is determined by the volumes of the solution being
extracted and the extracting solutions. Typical extractions in the laboratory are done in a separatory funnel,
while microscale extractions are done in a conical vial.
Extraction with organic solvents
Liquid-liquid extractions usually involve water and an organic solvent. Most common organic
solvents (diethyl ether, ethyl acetate, toluene, dichloromethane) are immiscible in water. If you place 50 mL
of ethyl acetate and 50 mL of water in a flask and stir the solution to mix it, you will not obtain a
homogeneous solution. Rather, if the solution is allowed to stand after stirring, two distinct liquid phases will
form in the flask: the more dense solvent as the lower layer and the less dense solvent as the upper layer.
Most organic solvents are much less polar than water. A general rule of thumb for solubility states
that “like dissolves like.” Polar compounds are more soluble in polar solvents than in nonpolar solvents, and
vice versa. The selective solubility of different compounds in polar versus nonpolar solvents allows the
separation of the compounds in a mixture by liquid-liquid extraction.
Suppose that we add compound X to a flask containing ethyl acetate and water, and stir the
contents of the flask to mix them. After mixing, the ethyl acetate and water will separate into two distinct
phases, and compound X will be found dissolved in both the ethyl acetate layer and in the water layer. How
compound X distributes between the two solvents is based on the solubility of X in each of the two solvents:
more of compound X will be found in the solvent in which it is more soluble. The ratio of the concentrations
of X in each of the immiscible solvents is called the distribution coefficient or the partition coefficient K
d
,
where:

12
The value of the distribution coefficient depends on the solubility of the compound in the two
solvents in the system. In the above system, if compound X has a higher solubility in ethyl acetate than in
water, at equilibrium the concentration of X in ethyl acetate will be greater than the concentration of
compound X in water, and the value of the distribution coefficient K
d
, will be greater than 1. If instead
compound X has a higher solubility in water than in ethyl acetate, at equilibrium the concentration of X in
water will be greater than the concentration of compound X in ethyl acetate, and the value of the distribution
coefficient K
d
will be less than 1.
The efficiency of a liquid-liquid extraction depends on the distribution coefficient of the desired
compound between the two solvents. If we want to extract an organic compound from an aqueous solution
into an organic solvent, it is desirable to use a solvent that has a much higher affinity for the compound than
does water. For example, at 25°C, the solubility of benzoic acid in water is 3.4 g per liter while the solubility
of benzoic acid in chloroform (CHCl
3
) is 222 g per liter. Water and chloroform are immiscible solvents. If a
solution of 1 g of benzoic acid in 400 mL of water is extracted with 400 mL of chloroform, we would expect
most of the benzoic acid to be transferred to the chloroform layer in which it is more soluble. The benzoic
acid will distribute itself between the two solvents in the ratio (approximately) of the solubilities in each
solvent:
No matter how much benzoic acid is present in the system, it will always be distributed between the
chloroform and water so that the ratio of the concentration in each solvent is 65.3.
From this estimate of the distribution coefficient, we can calculate how much benzoic acid is
present in the chloroform and water layers after the extraction. Let x = grams of benzoic acid in the water
layer and y = grams of benzoic acid in the chloroform layer.
Since we started with 1 g of benzoic acid, x + y = 1.
Using this equation along with the value for the distribution coefficient calculated above, we can
determine the concentration of benzoic acid in each layer. Or, since the volumes of both solvents are the
same:
The total amount of benzoic acid present is (x + y = 1). Rearranging this equation and substituting
in for the previous equation gives:

13
Solving this equation for y gives 0.015 g (15 mg) of benzoic acid in the water layer, and, since the
total amount of benzoic acid is 1 g, there is 0.985 g (985 mg) of benzoic acid in the chloroform layer.
Multiple extractions
In the previous example, one extraction with 400 mL of chloroform removed 98.5% of the benzoic
acid from the aqueous solution. If we divide the 400 mL of chloroform used in half and do two successive
extractions of the aqueous phase, the amount of benzoic acid extracted will increase.
The equation for the distribution coefficient for two 200 mL chloroform extractions of the 400 mL
aqueous solution of benzoic acid is:
In the first extraction, 1 g of benzoic acid is distributed between the phases, so (x + y) as before.
Solving the two equations in two unknowns gives x = 0.97 g in CHCl3 and y = 0.03 g in H2O. When the
aqueous phase is extracted a second time with a fresh 200 mL of chloroform, only 0.03 g of benzoic acid is
left in the aqueous phase to distribute between the two solvents. In this extraction the equation for the
distribution coefficient is the same but (x + y) = 0.03, and solving for x and y, the amount of benzoic acid in
each layer after the second extraction gives x = 0.0291 g in CHCl3 and y = 0.0009 g in H2O. Combining the
amounts of benzoic acid found in the two chloroform extracts gives 99.91% (0.9991 g of the original 1 g) of
the benzoic acid extracted into the chloroform layer by using two 200 mL extractions instead of 98.5%
removed with one 400 mL extraction. In general, it is always more efficient to carry out several extractions
using a small volume of solvent each time than to carry out a single extraction using a large volume of
solvent.
Acid-base extraction
Organic compounds are classified as being neutral, acidic, or basic depending on the types of
functional groups they contain. Many organic compounds, although just slightly polar overall, contain
functional groups that can act as a Bronsted-Lowry acid or base (i.e., they can donate or accept a proton,
respectively). Carboxylic acids, phenols, and thiols are examples of acidic functional groups; substituted
amines (including anilines) are examples of basic functional groups. Although the water-solubility of these
compounds is often limited because of their overall nonpolar character, their aqueous solubilities can be
dramatically increased through an acid-base neutralization reaction. This changes the compound into an
ionic salt that is very water soluble and will distribute almost completely into the aqueous layer.
Experiment 1 has been designed to illustrate how an acid-base extraction works.
Thin layer chromatography (TLC)
Chromatography: A General Introduction
Chromatography is the most versatile technique for separating mixtures. The technique derives its
name from early experiments in which plant pigments were separated into individual components such as

14
chlorophylls and xanthophylls by passing the mixture through a column packed with calcium carbonate.
Bands of different colors appeared on the column. Chromatography is not limited to colorful materials,
however.
Many different types or classes of chromatography exist and are used not only to separate, but also
to isolate and identify both in a qualitative and quantitative manner. What all forms of chromatography have
in common is that each employs a stationary phase and a mobile phase. Components of the mixture are
carried past the stationary phase by the flow of the mobile phase. The components distribute, or partition,
between the two phases, and separation occurs based on the average time a component spends in each of
the phases. For example, materials that are strongly attracted to the stationary phase will flow slowly;
whereas components that are highly soluble in the mobile phase will travel quickly. Different classifications
of chromatography are made based on the identity of each of the phases (for example, whether the mobile
phase is a gas or a liquid) and the operating conditions of the system.
Compounds partition between the two phases based on polarity. Adsorbents used for solid-liquid
chromatography are generally polar, so polar solutes adsorb strongly onto the stationary phase. They will
elute (move with the mobile phase) slowly. Nonpolar compounds are not as strongly adsorbed, and
therefore spend more time, on average, in the mobile phase and thus elute rapidly. For routine organic
laboratory work, silica gel (SiO
2
•nH
2
O) is the most common adsorbent.
Thin Layer Chromatography (TLC)
In TLC the adsorbent is spread in a thin layer over a solid support such as a sheet of aluminium (or
glass) to make a TLC plate. Samples are spotted at the bottom of the plate which is developed by placing it
in a chamber containing small amount of the mobile phase. The mobile phase (a solvent) wicks up the plate
by capillary action. TLC is used as an analytical technique, and provides qualitative information about a
sample. TLC is the most common and widely used method of analysis in a synthetic organic laboratory. It is
not, however, the most powerful technique for analysis. It is used because of low cost, rapid analysis time,
convenience, and simplicity.
As already mentioned, the most common stationary phase used is silica gel mixed with plaster of
Paris (calcium sulfate) to harden and support the adsorbent. You must handle TLC plates carefully and only
by the edges of the plates. The adsorbent coating is fragile and should remain clean prior to use.

15
The sample to be analyzed must be prepared as a dilute solution. The solvent chosen for this
should dissolve the compound well, and must evaporate rapidly. A small sample of this solution is spotted
near the bottom of a TLC plate with a fine glass capillary tube. The plate is typically prepared in advance by
drawing a light pencil line ~1 cm from the bottom of the plate, then making light cross marks to indicate
where the sample will be spotted. TLC is most often used to compare samples, so each plate will have
several spots. The plate is placed in a development chamber. Solvent moves up the plate by capillary
action. The plate is removed from the chamber when the solvent has risen to within one centimeter of the
top of the plate. A pencil line is immediately drawn across the top to indicate the height the solvent front
travelled.
The technical name for a substance being analyzed is “analyte”, but we rarely use that term. It will
be referred to as “the solute” (because it is in solution) or, more roughly, “the stuff you are analyzing,” or
“the stuff that you spotted,” or “the sample.” We will often relate molecular structure of the analyte to
different types of intermolecular forces. This will allow us to relate the structure of a molecule to a property
we can observe–the distance it travels along the plate in a TLC analysis. The mobile phase is also referred
to as the developing solvent, or “eluent” from the verb, “to elute”. Changing the developing solvent will
change the result of an analysis. Again, the types of intermolecular forces present are important and will be
related to how the mobile phase behaves.
Finally, YOU MUST DILUTE a sample before you can spot it on a TLC plate. A solution between
2% and 5% is usually sufficient. Place a few milligrams of the sample in a small disposable vial and add
approximately 0.5 mL of solvent. The solvent you use for this has no special name, and if selected
correctly, has no effect on the outcome of your TLC analysis. This solvent is simply used to dilute the
sample and must evaporate before the plate is developed. Select a solvent that dissolves a wide range of
samples and has a low boiling point. Diethyl ether or dichloromethane are often good choices.
Shown below are typical results from TLC analyses. These results illustrate the effect from
developing solvent polarity, and the effect of analyte polarity.

16
Effect of eluent polarity on TLC analysis
Effect of sample polarity on TLC analysis
TLC can be used to establish if two materials are definitely different (they travel different distances)
or possibly the same. Two different materials can coincidentally behave the same in TLC. Illustrated below
are some typical problems encountered in TLC.
Eluent polarity problems
Sample concentration problem

17
Experimental design problem
Visualization
Unless the materials being analyzed are colored (absorb visible light), a method for visualizing the
resulting spots is required. Several methods are available. Materials that absorb ultraviolet light are easily
visualized because the adsorbent has a fluorescent indicator added to it. When UV light at 254 nm is
shined on the plate, the adsorbent surface will fluoresce brightly and the compounds that absorb UV light
will be seen as dark spots. These spots may then be traced on the surface of the plate with a pencil.
Caution!
UV light is harmful. Minimize exposure to skin and do not shine in eyes.
This method is limited to materials that absorb UV light. Aromatic rings and conjugated ketones are
two important functional groups you will see in laboratory that absorb UV light.
The second method of visualization takes advantage of the fact that iodine will form a complex with
a wide variety of organic compounds. Place the plate to be visualized in a wide-mouth jar with iodine and
warm the bottom of the jar gently to fasten the process of sublimation. Yellow or brown spots will appear.
This method is temporary, so a permanent record should be made by tracing the outline of the spots on the
plate.
Other visualization reagents are available and are generally prepared as a solution that is sprayed
on the plate or in which the plate is dipped. Methods for preparing these reagents can be found in
handbooks of organic chemistry or chromatography.
Retention factor (R
f
)
Under a fixed set of conditions, a given compound always travels the same distance relative to the
distance the solvent front travels. This ratio of distances is called the retention factor and is abbreviated R
f
.
A sample calculation is shown below. It is customary to measure from the center of the spot.

18
Calculating a retention factor
The polarity of the mobile phase (eluent, developing solvent) also determines the speed at which
substances being analyzed will elute. As a general rule of thumb, any solute will elute more rapidly in a
more polar eluent. This is a rough guideline. The polarity guidelines are based on intramolecular forces
between adsorbent and solute, or adsorbent and solvent, including van der Waals force, dipole-dipole,
hydrogen bonding, coordination, and salt formation.
Recrystallization
The most common method of purifying solid organic compounds is by recrystallization. In this
technique, an impure solid compound is dissolved in a solvent and then allowed to slowly crystallize out as
the solution cools. As the compound crystallizes from the solution, the molecules of the other compounds
dissolved in solution are excluded from the growing crystal lattice, giving a pure solid.
Crystallization of a solid is not the same as precipitation of a solid. In crystallization, there is a slow,
selective formation of the crystal framework resulting in a pure compound. In precipitation, there is a rapid
formation of a solid from a solution that usually produces an amorphous solid containing many trapped
impurities within the solid's crystal framework. For this reason, experimental procedures that produce a
solid product by precipitation always include a final recrystallization step to give the pure compound.
The process of recrystallization relies on the property that for most compounds, as the temperature
of a solvent increases, the solubility of the compound in that solvent also increases. For example, much
more table sugar can be dissolved in very hot water (just below the boiling point) than in water at room
temperature. What will happen if a concentrated solution of hot water and sugar is allowed to cool to room
temperature? As the temperature of the solution decreases, the solubility of the sugar in the water also
decreases, and the sugar molecules will begin to crystallize out of the solution. (This is how rock candy is
made.) This is the basic process that goes on in the recrystallization of a solid.
The steps in the recrystallization of a compound are:
1. find a suitable solvent for the recrystallization;
2. dissolve the impure solid in a minimum volume of hot solvent;
3. use decolorizing carbon (if necessary);
4. remove any insoluble impurities by filtration;
5. slowly cool the hot solution to crystallize the desired compound from the solution;
19
6. filter the solution to isolate the purified solid compound.
1. Choosing a Solvent
The first consideration in purifying a solid by recrystallization is to find a suitable solvent. There are
four important properties that you should look for in a good solvent for recrystallization. First is that the
compound should be very soluble at the boiling point of the solvent and only sparingly soluble in the solvent
at room temperature.
This difference in solubility at hot versus cold temperatures is essential for the recrystallization
process. If the compound is insoluble in the chosen solvent at high temperatures, then it will not dissolve. It
the compound is very soluble in the solvent at room temperature, then getting the compound to crystallize
in pure form from solution is difficult. For example, water is an excellent solvent for the recrystallization of
benzoic acid. At 10°C only 2.1 g of benzoic acid dissolves in 1 liter of water, while at 95 °C the solubility is
68 g/L.
The second desirable property of a good recrystallization solvent is that the unwanted impurities
should be either very soluble in the solvent at room temperature or insoluble in the hot solvent. This way,
after the impure solid is dissolved in the hot solvent, any undissolved impurities can be removed by
filtration. After the solution cools and the desired compound crystallizes out, any remaining soluble
impurities will remain dissolved in the solvent.
A third important property of the recrystallization solvent is that it must not react with the compound
being purified. The desired compound may be lost during recrystallization if the solvent reacts with the
compound.
Finally, the recrystallization solvent should be volatile enough to be easily removed from the
compound after it has crystallized. This allows for easy and rapid drying of the solid compound after it has
been isolated from the solution.
Finding a solvent with the desired solubility properties is a search done by trial and error. First, test
the solubility of tiny samples of the compound in test tubes with a variety of different solvents (water,
ethanol, methanol, ethyl acetate, diethyl ether, hexane, toluene, etc.) at room temperature. If the compound
dissolves in the solvent at room temperature, then that solvent is unsuitable for recrystallization. If the
compound is insoluble in the solvent at room temperature, then the mixture is heated to the solvent's boiling
point to determine if the solid will dissolve at high temperature, and then cooled to see if it crystallizes from
the solution at room temperature.
2. Dissolving the Solid
Once a suitable solvent is selected, place the impure solid in an Erlenmeyer flask and add a small
volume of hot solvent to the flask. Erlenmeyer flasks are preferred over beakers for recrystallization
because the conical shape of an Erlenmeyer flask decreases the amount of solvent lost to evaporation
during heating, prevents the formation of a crust around the sides of the glass, and makes it easier to swirl
the hot solution while dissolving the solid without splashing it out of the flask.
Keep the solution in the Erlenmeyer flask warm on a hot plate or in a water bath, add small volumes
of hot solvent to the flask until the entire solid just dissolves. Swirl the solution between additions of solvent
20
and break up any lumps with a stirring rod or spatula. Occasionally there will be impurities present in the
solid that are insoluble in the chosen solvent even at high temperature. If subsequent additions of solvent to
the solution do not seem to dissolve any of the remaining solid, stop adding solvent to the solution (as this
will decrease the percent recovery of the desired compound) and filter the hot solution to remove the
insoluble impurities.
3. Use of Decolorizing Carbon
Colored impurities are sometimes difficult to remove from solid mixtures. These colored impurities,
often due to the presence of polar or polymeric compounds, can cause a colorless organic solid to have a
tint of color even after recrystallization. Decolorizing or activated carbon is used to remove the colored
impurities from the sample. Decolorizing carbon is very finely divided carbon that provides high surface
area to adsorb the colored impurities.
Very little decolorizing carbon is needed to remove the colored impurities from a solution. You must
be judicious in your use of decolorizing carbon; if too much is used, it can adsorb the desired compound
from the solution as well as the colored impurities. After the impure solid sample is dissolved in hot solvent,
a small amount of decolorizing carbon, about the size of a pea, is added to the hot solution. This must be
done carefully to avoid a surge of boiling activity from the hot solution (it is like adding a zillion little boiling
stones). The solution is stirred and heated for a few minutes and then filtered hot to remove the
decolorizing carbon. The resulting filtrate should be colorless and the recrystallization process continues as
before.
4. Removing Insoluble Impurities by Hot Filtration
A hot filtration is a gravity filtration using a stemless (or very shortstemmed) funnel (see Figure
below) that is done on a hot plate to keep the solution and the apparatus warm during the filtration. This
prevents the dissolved solid from crystallizing out of the solution prematurely. Place the funnel, lined with a
piece of folded or fluted filter paper, on an Erlenmeyer flask that contains a few milliliters of the
recrystallization solvent. Heat the flask until the solvent boils and allows the vapors to warm the filter
apparatus before beginning the filtration. Before filtering, it is a good idea to add a small amount (10-20%)
of extra solvent to keep the solid from crystallizing out during filtration. While the hot filtration apparatus is
still on the hot plate, rapidly filter the hot solution. After the solution has been filtered, wash the residue on
the filter paper with a small volume of fresh, hot solvent to dissolve any compound that may have
precipitated during the filtration.
Any insoluble impurities are removed while the solution is still hot, before the desired compound
begins to crystallize from the solution. These impurities are removed by hot filtration, a filtering technique
that requires certain precautions to prevent the solute from crystallizing out during the procedure.

21
Hot filtration
5. Crystallizing the solid
After the insoluble impurities have been removed, cover the flask containing the hot filtrate with a
watch glass and set it aside undisturbed to cool slowly to room temperature. As the solution cools, the
solubility of the dissolved compound will decrease and the solid will begin to crystallize from the solution.
After the flask has cooled to room temperature, it may be placed in an ice bath to increase the yield of solid.
Do not rapidly cool the hot solution by placing the flask in an ice bath before it has cooled to room
temperature. This will result in a rapid precipitation of the solid in an impure form because of trapped
impurities.
Sometimes the dissolved compound fails to crystallize from the solution on cooling. If this happens,
crystallization can be induced by various methods. One way to induce crystallization is by scratching the
inner wall of the Erlenmeyer flask with a glass stirring rod. This is believed to release very small particles of
glass which act as nuclei for crystal growth. Another method of inducing crystallization is to add a small
crystal of the desired compound, called a seed crystal, to the solution. Again, this seed crystal acts as a
template on which the dissolved solid will begin crystallizing. If neither of these two techniques results in
crystallization, the compound was probably dissolved in too much hot solvent. If you believe that you may
have too much solvent for the amount of dissolved compound, reheat the solution to boiling, boil off or distill
some of the solvent, and then allow the solution to cool to room temperature again to effect crystallization.
6. Isolating the Solid by Suction Filtration
Once the compound has completely precipitated from the solution, it is separated from the
remaining solution (also called the mother liquor) by filtration. Typically this is done by vacuum or suction
filtration (Figure) using either a Büchner funnel or a Hirsch funnel. Line the bottom of the Büchner or Hirsch
funnel with a piece of filter paper that is large enough to cover the holes in the bottom plate of the funnel
without curling up on the sides of the funnel. Place a neoprene adapter on the stem of the funnel and insert
it in the top of a Kitasato-flask (a thick-walled Erlenmeyer flask with a side-arm) that has been securely
clamped to a ring stand.
Using a piece of thick-walled vacuum tubing, connect the side-arm of the filter flask to a vacuum
pump. Turn the pump on force to create a vacuum through the system. If necessary, carefully adjust the

22
piece of filter paper so that it covers all of the holes in the funnel, and then dampen it with a small volume of
cold solvent; this will create a better seal between the filter paper and the plate in the funnel, preventing any
solid from getting under the filter paper and passing through the funnel. Slowly pour the recrystallization
solution into the funnel and allow the suction to pull the mother liquor through. Rinse the Erlenmeyer flask
with a small volume of cold recrystallization solvent to remove any remaining solid. Add this solvent to the
funnel and then wash the solid in the funnel, called the filter cake, with a few milliliters of fresh, cold
recrystallization solvent to remove any remaining mother liquor and dissolved impurities.
Vacuum filtration
Leave the aspirator on for a few minutes and allow air to pass through the crystals to dry them.
After pulling air through the crystals for a brief time, remove the vacuum from the system by disconnecting
the vacuum tubing from the aspirator before turning the water off. If you turn the pump off first, oil can be
sucked into the filter flask and may contaminate the product. The filter cake is removed from the funnel by
carefully prying it from the filter using a spatula. The cake of crystals will still be slightly wet with solvent and
should be allowed to dry thoroughly before measuring the weight or melting point.

23
Common organic solvents
SOLVENT Boiling point (ºC) Specific gravity
acetic acid 118 1.05
acetone 56 0.79
chloroform 61 1.48
cyclohexane 81 0.78
dichloromethane 40 1.32
ethanol 78 0.80
ether (diethyl) 35 0.71
ethyl acetate 77 0.90
hexane 69 0.66
methanol 65 0.79
2-propanol 82 0.79
tetrahydrofuran 65 0.99
toluene 111 0.87
m-xylene 139 0.87

24
Experiment 1.- Using Extraction and NMR to isolate and identify the
analgesic active principles of a sample
Overview: Analgesics
The medicinal effects of willow bark extracts have been
known for centuries. Hippocrates used a willow bark preparation to
alleviate pains and reduce fever. Early in the nineteenth century it
was recognized that the active principle in willow bark extracts, as
well as in extracts from flowers of the meadowsweet plant, was
salicylic acid. It was found that salicylic acid was a very effective
analgesic, antipyretic, and anti-inflammatory agent. It was also
found that salicylic acid had undesirable side effects: gastrointestinal irritation.
Felix Hoffmann, a chemist at the Bayer Company in Germany, discovered in
1893 that a rather simple chemical modification of salicylic acid afforded a new compound that
not only had superior pain-relief power than salicylic acid but also none of its side effects. The
newly synthesized compound, acetylsalicylic acid, was clinically evaluated and its industrial
production began in 1899 under the trademark of Bayer Aspirin (“a” from acetyl and “spirin”
from the Latin name for the meadowsweet plant, spirea). Aspirin is probably the most versatile
drug ever devised. Besides the effects mentioned above, it has been shown that aspirin reduces the
risk of heart attack in patients with a history of cardiovascular disease.
Other analgesics have been developed in the last fifty years; among them naproxen (the active
ingredient in Aleve), ibuprofen (the active ingredient in Advil), and especially acetaminophen
(the active ingredient in Tylenol) have gained wide acceptance.
Naproxen
Ibuprofen
Acetaminophen (paracetamol)
Acetylsalicylic acid
The pharmaceutical compositions contain additional components. A very good example is
caffeine, a ubiquitous substance. From a historical standpoint, caffeine is one of the oldest
substances consumed by man in the form of
coffee beans, teas, guanara beans and mate.
Caffeine is commonly found in many
analgesic products as an adjuvant. Caffeine is
almost completely absorbed in the
gastrointestinal tract. Since caffeine is lipid soluble, it is rapidly absorbed into the brain where it
becomes active within 6–8 min. Caffeine has some analgesic properties itself and because of its
Meadowsweet
plant
Willow bark
Salicylic acid
Caffeine

25
rapid absorption and distribution, is useful as an adjuvant in combination with analgesic
medications.
Overview: The molecules
Aspirin has two functional groups attached to a benzene ring: an ester group, which is
neutral, and a carboxylic acid group with acidic character.
Ibuprofen has carboxylic and benzene groups as well and a more complicated carbon
framework.
Naproxen bears a carboxylic acid too, a neutral ether function and a naphthalene aromatic
core.
Aspirin, Ibuprofen and Naproxen has the carboxylic group in common that makes them
acidic compounds with pK
a
values of 3.5, 5.2 and 4.2, respectively. The ester (aspirin) and ether
(naproxen) functions are neutral. The aromatic nucleus of these molecules is highly hydrophobic
and, as a consequence, they are very insoluble in neutral aqueous solutions. However, they
dissolve in basic solutions because of the carboxylic group.
The conjugate bases of aspirin, ibuprofen or naproxen are salts and thus water soluble;
therefore, they can be removed from the organic solvent layer by extraction with a weak aqueous
base (sodium bicarbonate). Re-acidification of this basic aqueous layer will regenerate the organic
acid, which will precipitate from the aqueous solution due to the limited solubility in water of the
acids.
Acetaminophen (paracetamol) is a phenol (pK
a
~ 10) with a neutral acetamido group in
para position. Acetaminophen is a very weak acid, about six orders of magnitude less acidic than
aspirin. It dissolves in strong basic solutions like sodium hydroxide (pH > 11) but it does not
dissolve under mild basic conditions (pH = 8-9). Additionally, the solubility of paracetamol in
organic solvents is much lower than that of aspirin, ibuprofen and naproxen.
Caffeine, contrary to the previous compounds, is rather soluble in water (ca. 2 g/100 mL at
25ºC). Yet, although a weak one, caffeine is a base and, consequently, its solubility in water is
even increased in the presence of strong acids.
Because aspirin, ibuprofen and naproxen are more acidic than acetaminophen, in principle,
the latter chemical could be separated by acid-base extraction using a weak base such as sodium
bicarbonate. Sodium bicarbonate reacts with the carboxylic acid function forming a sodium salt,
but it does not react with the weaker acidic phenol group of acetaminophen. However, their
separation can be accomplished in a much easier way because of the unusual solubility behavior of
acetaminophen. This compound is only slightly soluble in organic solvents such as methylene
26
chloride and tert-butyl methyl ether, whereas aspirin, ibuprofen and naproxen are readily soluble
in these solvents.
Overview: The experiment
Your main task is to determine which active principle(s) of those described above is(are)
contained in the sample given to you. To accomplish the task you will perform in this experiment
the systematic separation of all components of the given sample carrying out a complete extraction
procedure with bases (sodium bicarbonate and sodium hydroxide) and hydrochloric acid. NMR
spectra of all of your isolated species will be recorded and compared to spectra of the real
components.
Your sample will be first dissolved in methylene chloride. This solvent does not dissolve
acetaminophen and in the case that this component is present, it can be easily collected by simple
filtration. Then the organic solution will be sequentially extracted with sodium bicarbonate,
sodium hydroxide and hydrochloric acid.
The Figure below shows two alternative routes to perform the separation. The order of acid
and base extraction can be permuted. Therefore, it is your decision which route has to be taken
(HINT: Deem caffeine solubility in water!). CAUTION: The bases must always be used in the
indicated order; sodium bicarbonate first, sodium hydroxide second.
The bicarbonate extracts, once they have been carefully neutralized with hydrochloric acid,
would render the stronger acids (carboxylic acids) whereas the sodium hydroxide extraction, after
the corresponding neutralization, would yield the weaker acids (phenols). At this point you must
be careful at assessing whether the extraction of the strong acids with sodium bicarbonate was
complete or not. Otherwise, the remaining strong acids, if you left any, will be extracted by
sodium hydroxide and the separation would not be satisfactory. That is the reason why the order of
the bases cannot be permuted.
Between the base and acid extraction is convenient to wash the organic phase with a small
amount of brine. The acid extracts, conveniently neutralized with sodium hydroxide, would render
the bases present in your sample.
You will assess the success of the separation by comparing the NMR spectra recorded from
all your fractions with the spectra of the real compounds.

26
Strategy 1
Strategy 2
Figure. Flowcharts for the separation of the components of a sample problem.

28
Safety First
HCl and NaOH are corrosive and toxic. HCl is highly toxic. Handle them
with care.
Methylene chloride (dichloromethane, DCM) is a possible carcinogen. Handle them in a
well-ventilated place.
Experimental Procedure
1. Weigh the given sample (weight should be close to 1 g). Place the sample in a 100 mL
Erlenmeyer flask and add 50 mL of DCM. Stir for a while trying to dissolve as much sample
as possible.
2. Should a suspension be formed, filter the solid using a simple funnel and a
pleated filter. Keep the filter in a safe place and, once it is dry, transfer the
solid to a weighed vial in order to analyze its contents later on.
3. Place the DCM solution in a separating funnel.
IMPORTANT: At this moment you would already have to have it clear which of the two
extraction strategies you are going to follow (HINT: Deem caffeine solubility in water!). To
follow the Strategy 1 flowchart (base extraction first) just go on. Otherwise (Strategy 2
flowchart; acid extraction first) perform now steps 12-14 first, then carry out steps 10-11 and
finally steps 4-9.
4. Extract the organic layer with 10 mL portions of saturated aqueous solution of sodium
bicarbonate.
(Your sample may not be colored)
5. Transfer the DCM layer (bottom) to the Erlenmeyer where you dissolved the sample and the
aqueous layer to a 50 mL Erlenmeyer flask. Use 10% HCl to carefully neutralize (control pH
with pH paper) the aqueous phase. Caution: strong effervescence will take place. Do not lose
any product. Look at the possible formation of a precipitate when the solution is acidic.

29
Transfer the aqueous phase to a test tube labeled as SB1N (after Sodium Bicarbonate,
extraction 1, Neutralized).
6. Repeat steps 3 and 4 until no more precipitate forms on neutralization. Keep the test tubes
labeled SBnN for ulterior filtration.
7. Transfer the DCM layer again to the separation funnel. Extract the organic layer with 10 mL
portions of 10% aqueous solution of sodium hydroxide.
8. Transfer the DCM layer (bottom) to the Erlenmeyer where you dissolved the sample and the
aqueous layer to a 50 mL Erlenmeyer flask. Use 10% HCl to carefully neutralize (control pH
with pH paper) the aqueous phase. Caution: heat develops. Do the neutralization in an ice
bath. Look at the possible formation of a precipitate when the solution is acidic. Transfer the
aqueous phase to a test tube labeled as SH1N (after Sodium Hydroxide, extraction 1,
Neutralized).
9. Repeat steps 6 and 7 until no more precipitate forms on neutralization. Keep the test tubes
labeled SHnN for ulterior filtration.
10. Transfer again the DCM layer to the separation funnel. Extract the organic layer with 10 mL
portions of brine.
11. Transfer the DCM layer (bottom) to the Erlenmeyer where you dissolved the sample and the
aqueous layer to a test tube labeled brine. Keep it and discard when the experiment is finished.
12. Transfer the DCM layer again to the separation funnel. Extract the organic layer with 10 mL
portions of 10% hydrochloric acid.
13. Transfer the DCM layer (bottom) to the Erlenmeyer where you dissolved the sample and the
aqueous layer to a 50 mL Erlenmeyer flask. Use 10% NaOH to carefully neutralize (control
pH with pH paper) the aqueous phase. Caution: heat develops. Do the neutralization in an ice
bath. Look at the possible formation of a precipitate when the solution is basic. Transfer the
aqueous phase to a test tube labeled as HA1N (after Hydrochloric Acid, extraction 1,
Neutralized).
14. Repeat steps 11 and 12 until no more precipitate forms on neutralization. Keep the test tubes
labeled SHnN for ulterior filtration.
15. Dry the organic layer with a small amount of solid anhydrous magnesium or sodium sulfate.
Filter the solid off on a pleated filter and keep the filtrate for ulterior evaporation of the DCM.

30
16. Filter the test tubes labeled SBnN using a small Buchner filter, a
Kitasato and vacuum. Leave the air current passing for a while
through the filter until it is dry. Carefully collect the solid in a
pre-weighed vial labeled as strong acids. Weigh its contents.
17. Clean the Buchner and Kitasato and filter the test tubes labeled
SHnN using the previous procedure. Carefully collect the solid
in a pre-weighed vial labeled as weak acids. Weigh its contents.
18. Clean the Buchner and Kitasato and filter the test tubes labeled HAnN using the previous
procedure. Carefully collect the solid in a pre-weighed vial labeled as bases. Weigh its
contents.
19. Place the organic layer of step 14 in a pre-weighed round-bottomed
flask with B-29 ground-glass socket and evaporate the solvent using a
rotary evaporator. Once the solvent has been removed, weigh the flask
and label it as neutral residue.
NMR spectra of the possible components
9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
299.39101.31100.00
No. (ppm) Value
1 [2.25 .. 2.38] 299.394
2 [7.10 .. 7.23] 98.288
3 [7.32 .. 7.44] 101.307
4 [7.54 .. 7.72] 99.630
5 [7.96 .. 8.13] 100.000
8.1 8.0 7.9 7.8 7.7 7.6 7.5 7.4 7.3 7.2 7.1 7.0
Chemical Shift (ppm)
102.11100.00 99.63 98.29
Chloroform-d
O
OH
O
OCH
3
1
H NMR Spectrum of Aspirin (300 MHz, CDCl
3
)

31
9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
600.00398.61 302.44200.11 99.9098.44
Methanol-d4
No. (ppm) Value
1 [0.74 .. 0.99] 600.000
2 [1.29 .. 1.52] 302.438
3 [1.69 .. 1.95] 99.895
4 [2.31 .. 2.54] 200.105
5 [3.53 .. 3.77] 98.440
6 [6.93 .. 7.32] 398.611
7.25 7.00
Chemical Shift ...
398.61
3.75 3.50
Chemical Shift (p...
98.44
2.5 2.4 2.3
Chemical Shi...
200.11
1.9 1.8 1.7
Chemical Shift (ppm)
99.90
1.5 1.4 1.3
Chemical Sh...
302.44
0.9 0.8
Chemical Shif...
600.00
CH
3
OH
O
CH
3
CH
3
1
H NMR Spectrum of Ibuprofen (300 MHz, CD
3
OD)
9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
379.62 300.00189.82185.20
Chloroform-d
No. (ppm) Value
1 [1.38 .. 1.56] 300.000
2 [3.69 .. 3.93] 379.620
3 [6.93 .. 7.12] 189.821
4 [7.54 .. 7.75] 185.201
5 [7.80 .. 8.00] 184.850
CH
3
O
OH
O
CH
3
8.00 7.75 7.50 7.25 7.00
Chemical Shift (ppm)
189.82185.20184.85
Chloroform-d
3.85 3.80 3.75
Chemical Shift (ppm)
379.62
1.500 1.450
Chemical S...
300.00
1
H NMR Spectrum of Naproxen (300 MHz, CDCl
3
)
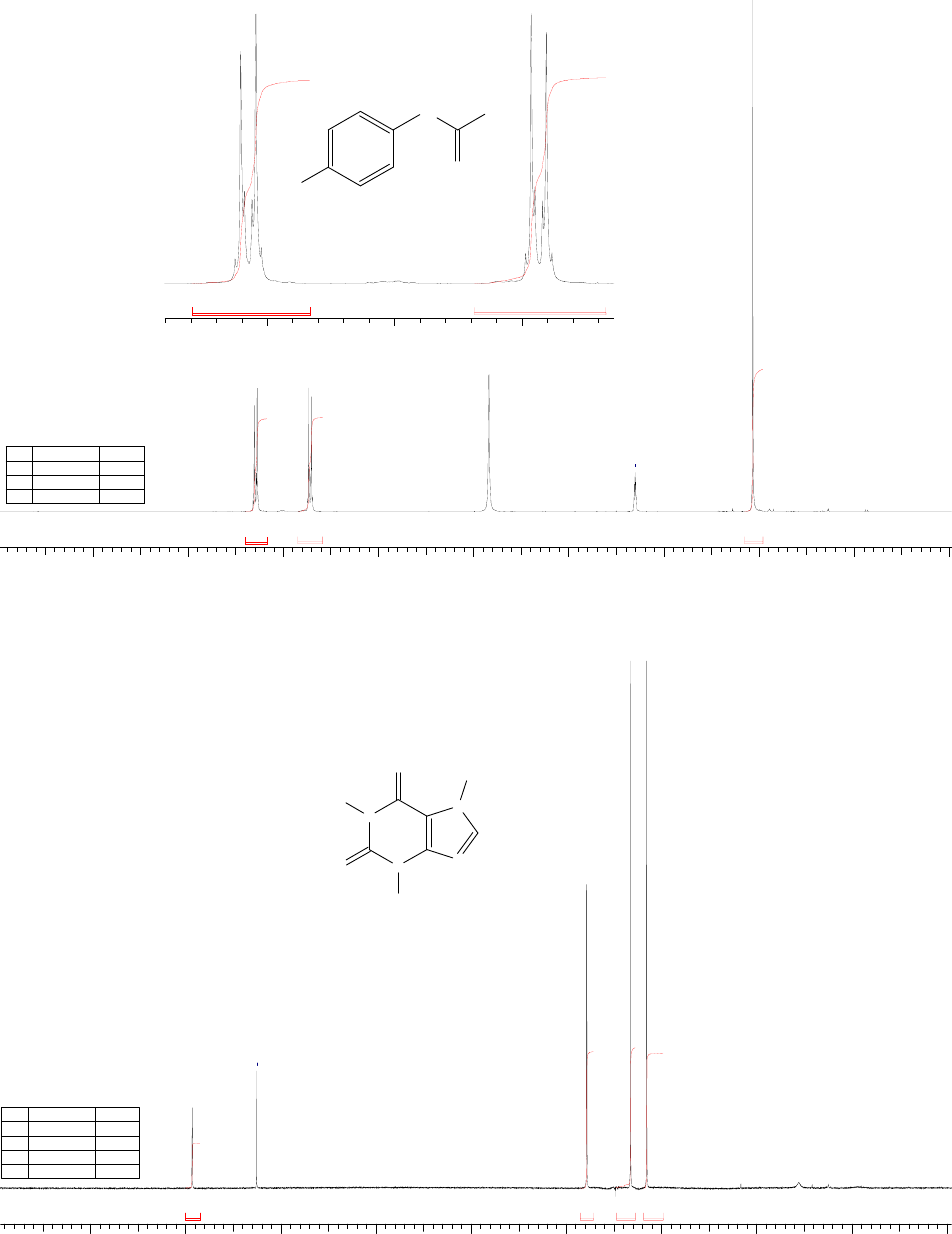
32
9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
300.00198.27195.59
Methanol-d4
No. (ppm) Value
1 [1.96 .. 2.15] 300.000
2 [6.58 .. 6.84] 198.267
3 [7.17 .. 7.40] 195.590
7.25 7.00 6.75
Chemical Shift (ppm)
198.27195.59
NH
OH
O
CH
3
1
H NMR Spectrum of Paracetamol (300 MHz, CD
3
OD)
No. (ppm) Value
1 [3.14 .. 3.26] 345.963
2 [3.30 .. 3.44] 342.479
3 [3.77 .. 3.90] 342.599
4 [7.90 .. 8.06] 100.000
N
N
N
N
O
O
CH
3
CH
3
CH
3
9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
31.5930.83 30.2710.00
Chloroform-d
1
H NMR Spectrum of Caffein (300 MHz, CDCl
3
)

33
TLC Analysis
In this experiment you may use Thin Layer Chromatography (TLC) to determine the
composition of your sample. You will be given two silica-gel TLC plates where four standards
(aspirin, ibuprofen, acetaminophen and caffeine), your sample (you had to have kept a small
amount of it) and the extraction fractions will be separately spotted. The standard compounds will
be available as solutions of ca. 1 g of each substance dissolved in 20 mL of a 1:1 mixture of DCM
and ethanol. You have to dissolve on your own your sample and the extraction fractions in the
same solvent mixture at a concentration similar to that of the standards. The purpose of the first
plate (standards plus sample) is to determine which standards are present in the sample by means
of your comparing the various retention factors (R
f
). The second plate (sample plus extraction
fractions) will serve you to assess your extraction skills.
It is possible to use several developing solvents (eluents) for this experiment but ethyl
acetate containing 0.5% glacial acetic acid is preferred. The small amount of acid supplies protons
and suppresses ionization of aspirin and ibuprofen, allowing them to travel upward on the plates in
their protonated form. Without the acid these compounds do not move.
Two methods of visualization will be used to observe the positions of the spots on the developed
TLC plates. First, the plates will be observed while under illumination from a short-wavelength
ultraviolet lamp. Under these conditions, some of the spots will appear as dark areas on the plate,
whereas others will fluoresce brightly. This difference in appearance under UV illumination will
help you to distinguish the substances from one another. You will find it very convenient to lightly
outline the spots with a pencil and place a small “x” on the fluorescent spots. Iodine vapors will be
used as a second method of visualization. Not all the spots will be revealed but some will turn
yellow, tan or brown. The difference in the behaviors of the various spots can be used to further
differentiate among them.
Please, read the provided laboratory manual for further guidance as to the correct
performance of the TLC experiment.
Report
Pre-lab
1. Make a table showing the physical properties (molecular mass, m.p., b.p. solubility,
flammability [for solvents only], and toxicity/hazards) of aspirin, ibuprofen, naproxen,
acetaminophen (synonym: N-acetyl-4-hydroxyaniline), caffeine, acetone, methylene chloride,
and ethyl acetate.
2. Classify the following compounds as acidic, basic, or neutral: acetaminophen, aspirin,
ibuprofen, naproxen, and caffeine. Identify the functional groups present in each molecule and
determine which ones affect their acid-base properties.
3. Could you use NaOH to separate aspirin and acetaminophen by acid-base extraction? Briefly
justify your answer.
4. Study the structure of caffeine and decide whether all nitrogen atoms are basic or not. Why?

34
In-lab
1. Calculate the yields of the separated components using the starting weight of the whole sample.
2. Report the melting points and compare them with literature values.
3. Report and discuss the NMR results.
4. Did you identify the sample components?
5. Was the separation effective?
6. Did you find any unknown component?
Post-lab questions:
1. Provide an IUPAC name for aspirin, ibuprofen, acetaminophen and naproxen (use the internet
and Wikipedia but, please, do
try to understand how the names are made up).
2. All analgesic compounds and caffeine are wildly soluble in ethanol. Could you have used
ethanol instead of dichloromethane to extract the caffeine from tea? Why or why not? You can
use ethyl acetate instead of dichloromethane for the extraction. What modification of the
procedure related to density would be necessary?
3. Aspirin is soluble in organic solvents such CH
2
Cl
2
or EtOAc, but can also be dissolved in dilute
aqueous NaOH. Explain why and write any possible reaction involved.
4. What is the purpose of adding sodium bicarbonate to the flask in step 4? Be specific and show a
chemical reaction relevant to the question.
5. What can you do if you do not know which layer is which in an extraction procedure?
6. What is the purpose of washing the organic layer with saturated aqueous sodium chloride
(otherwise known as brine)?
7. A student measured a melting point of the isolated caffeine to be 210-215°C (literature value is
236°C)? Give a simple explanation for this result.
8. At 25 °C, 1.0 g of caffeine will dissolve either in 47 mL of water, or in 8.1 mL of chloroform,
or in 86 mL of benzene, or in 370 mL of diethyl ether. Calculate the solubility (in grams/100
mL) of caffeine in these four solvents. Calculate the partition coefficient of caffeine between
water and each of the other three solvents. Which of these three organic solvents would be the
best choice for extracting caffeine from an aqueous solution?
9. Small portions of naphthalene and 2-naphthol (look up the chemical formulae in
internet) are dissolved in hexane. A dilute aqueous solution of NaHCO
3
is added and
the mixture is then shaken vigorously. A small portion of the top layer is analyzed by
TLC. The result is shown on the indicated plate. What information does the TLC
analysis show regarding this attempt to separate the two substances? Provide an
explanation.
10. Outline a flow chart to show how you could separate a mixture of the following
compounds by using acid-base extraction. All three compounds are crystalline solids and soluble
in common organic solvents.

35
Experiment 2.- Preparation of an azoic dye: Para-red
Overview: Dyes
A dye or colorant is a substance capable of permanently staining animal or vegetal fibers.
Dyes or colorants have been used from ancient times and extracted from some plants (turmeric,
indigo, etc), animals (woodlouse, mollusks, etc) or minerals. In present times, the chemical
industry has developed synthetic dyes in order to have a complete color palette and comply with
stringent conditions like sanitary ones, stability towards light and high resilience upon repeated
washings.
Dyes can be classified in terms of the organic functional group that characterizes their
structure. Among them the azoic dyes stand out. The azo group is a functional group consisting of
a double bond sustained between nitrogen atoms, with each nitrogen atom attached to another
group: R-N=N-R’. When R and R’ are aromatic rings and the N=N bond is thus conjugated with
their systems, the compound strongly absorbs visible light and hence displays intense colors.
Overview: The azo compounds
The azo group results from the reaction between a diazonium salt and an aromatic ring as
shown in the Scheme. In turn, the diazonium salt is formed by the action of nitrous acid on an
aromatic amine as follows:
NN
R
1
R
2
+
R
2
N
N
R
1
Diazonium salt
Azo derivative
NH
2
R
1
NaNO
2
H
Overview: Aromatic substitution
The first reaction to be performed is the introduction of a nitro group (nitration) on the
aromatic ring of anilide. This is an example of a very important category of organic reactions:
aromatic substitution.
The amide group of anilide is an electron donor as it may be deduced from the following
resonance forms.
HN
O
HN
O
HN
O
HN
O

36
As a consequence, the aromatic ring has an excess of electronic density at positions 2 and 6
(ortho) and 4 (para). Any electrophilic species would attack the ring at position 4 predominantly
because in addition to being electronically favorable, it is the least sterically hindered one.
In the particular case of the experiment, the electrophile is [NO
2
+
], obtained from the
mixture of concentrated nitric and sulfuric acids. The latter is stronger and actually makes nitric
acid to act as a base leading to [NO
2
+
] that in situ attacks the aromatic ring of acetanilide.
N
O
OHO
H
2
SO
4
N
O
OHHO
HSO
4
H
2
O
ONO
HSO
4
HN
O
ONO
HN
O
NO
2
HSO
4
H
2
SO
4
HN
O
NO
2
MAJOR
MINOR
+
In summary, you will carry out a multistep synthesis combined with the use of separation
(chromatography) and identification techniques (NMR).
Safety First
Concentrated sulfuric acid is very corrosive. It may cause severe burns.
Avoid skin contact.
Concentrated nitric acid is very corrosive. It may cause severe yellowish burns. Avoid skin
contact.
Concentrated hydrochloric acid is very corrosive. It may cause severe burns. Avoid skin
contact. Avoid breathing the fumes.
The sulfo-nitric mixture must be pre-formed in the hood and care must be extreme in
avoiding spills.

37
Experimental procedures
Procedure 1 (VIDEO: Steps 1 & 2): Preparation of nitroacetanilide from anilide
This reaction is actually performed in two steps as follows:
HN
O
HN
O
NO
2
H
2
SO
4
HN
O
NO
2
MAJOR
MINOR
+
HNO
3
1) conc. HCl
NH
2
NO
2
NH
2
NO
2
+
2) NH
4
OH
MAJOR
MINOR
The first step involves nitration of acetanilide with the sulfuric-nitric mixture and the
second one the acid hydrolysis with hydrochloric acid of the intervening nitroacetanilide followed
by neutralization with ammonium hydroxide.
Procedure 2 (VIDEO: Step 3): “Coupling” reaction
It involves the generation of the diazonium salt by treatment of 4-nitroaniline with sodium
nitrite and its “coupling” with the appropriate aromatic compound, in this particular case 2-
naphtol, a phenol derived from naphthalene. The following Scheme shows the pertinent reactions:
NH
2
NO
2
NaNO
2
H
2
SO
4
N
NO
2
N
N
NO
2
N
OH
H
NO
2
N
N
HO
Para-red dye
2-Naphtol
Diazonium salt
Summary of materials and reagents
Materials and Reagents Amount (weight / volume)
Acetanilide 5.4 g
Concentrated H
2
SO
4
14.6 mL
Concentrated HNO
3
3 mL
Concentrated HCl 28 mL
4-nitroaniline (from procedure 1) 1.4 g
Sodium nitrite 0.75 g
2-naphtol 1.5 g
2N NaOH 10 mL

38
4-Nitroanilide preparation (Step 1)
Prepare in the hood the sulfuric-nitric mixture by carefully mixing 2.6 mL of 97% sulfuric
acid (d = 1.84 g/mL) and 3 mL of 69% nitric acid (d = 1.41 g/mL) and keep it in a safe place.
Place 12 mL of concentrated sulfuric acid in an Erlenmeyer flask containing a magnetic bar and
cooled in a water-ice bath. While vigorously stirring with the aid of a magnetic plate, add
acetanilide (5.4 g, 40 mmol) in small portions with the help of a spatula. Then add the sulfuric-
nitric mixture very slowly not letting the temperature raise above 35 ºC (use a thermometer). Once
the addition is complete, remove the reaction flask from the ice-water bath and continue the
vigorous stirring at room temperature for 1 h. The Erlenmeyer contents are then poured over 250
mL of crushed ice contained in a beaker. A precipitate should be clearly formed that is filtered off
with the help of a Büchner funnel, a kitasato flask and vacuum. The solid collected in the Büchner
funnel is washed several times with cold water until the washings are neutral to pH paper. The
solid is dried as much as possible in the Büchner funnel under air flow. After ca. 30 min of drying,
take a small sample of the solid (300-400 mg) in a vial (see NOTE) and submit the rest of the
crude product to the hydrolysis conditions of the next procedure.
While the hydrolysis proceeds, analyze a small portion of the sample by thin layer
chromatography (TLC; eluant ethyl acetate/hexane) in order to qualitatively investigate its
composition. An NMR spectrum of the sample will be recorded as well (see the annex to this
practice). Using the rest of the sample (ca. 300 mg) you should carry out column chromatography
on silica-gel (see general procedure). Collect the necessary fractions and analyze them by TLC.
Mix the fractions containing the same component, evaporate the solvent and calculate the yields of
2- and 4-nitroanilide.
NOTE
: You must get rid of the water this sample contains in order to carry out chromatography
with it. Dissolve the sample in the necessary amount of ethyl acetate and add solid, anhydrous
sodium or magnesium sulfate until you observe part of the solid loose. Filter the solid off with a
pleated filter (not under vacuum) and evaporate the solvent of the filtrate. You should get the solid
product back reasonably free of water.
4-Nitroaniline preparation (Step 2)
The solid filtered from the previous procedure is transferred to a 250 mL round-bottomed
flask bearing a magnetic bar. Water (80 mL) is added and stirring is started until an homogeneous
paste is produced. Concentrated hydrochloric acid (36%, 28 mL) is then carefully added and the
mixture is heated under reflux for 30-35 min. The flask is allowed to cool in a cold water bath and
the reaction mixture is poured over 200 mL of crushed ice. Ammonium hydroxide is added until

39
the solution turns basic to pH paper. The precipitated solid is filtered under vacuum, washed with
water, dried and weighed (m.p. = 146 ºC for 4-nitroaniline). Estimate the overall yield with respect
to the starting anilide.
Para-red dye preparation (Step 3)
The diazonium salt of 4-nitroaniline is prepared by dropwise addition of a sodium nitrite
solution (0.75 g in 2 mL of water) over a stirred solution of the previously obtained 4-nitroaniline
in 5 mL of 50% sulfuric acid, contained in a 25 mL Erlenmeyer flask cooled in an ice/salt bath. It
is very important the addition to be performed at such a rate that the reaction temperature is
scrupulously maintained in the 0-5 ºC interval. The produced cold solution of the diazonium salt is
then poured over a solution of 2-naphtol in 10 mL of 2N sodium hydroxide solution. The pH must
be tested for basicity, otherwise some more sodium hydroxide solution must be added until clearly
basic. The red dye precipitates in the reaction medium. Some solid sodium chloride could be added
to the solution to complete precipitation. The solid is then filtered off under vacuum and dried
(m.p. = 246-248 ºC for para-red dye). Estimate the yield of this step and the overall yield from the
starting anilide.
Column Chromatography (general procedure)
Column chromatography (CC) is used for the separation of mixtures and the purification of
substances at preparative (not analytical) scale. The foundations of CC are the same as those
explained for TLC. The usual stationary phase in both cases is silica-gel. In CC the silica-gel is a
powder of a given particle size (mesh) that is placed in a glass column of a given diameter bearing
a stopcock or valve at the bottom part and a reservoir at the top.
First a low viscosity solvent
system (e.g., ethyl acetate/hexane
mixture) has to be found which
separates the mixture and moves the
desired component on analytical
TLC to an R
f
of ca. 0.35. If several
compounds running very close each
other on TLC are to be separated,
adjust the solvent to put the midpoint between the components at R
f
= 0.35. If the separation of the

40
compounds is larger, adjust the R
f
of the less mobile component to 0.35. Having chosen the
solvent, a column of the appropriate diameter (see Table I) is selected.
The following steps must be complied when running a CC (watch the video):
Filling the column with the stationary phase:
1. Place a small cotton plug at the bottom of the column (A). Use a long enough piece of glass
tubing to help hold it in the bottom narrowing part of the column. The plug must not be
either too loose or too tight.
2. Firmly attach the column to a vertical stand with the necessary clamps.
3. Introduce 1 cm height of sea sand right above the cotton plug.
4. Place the right amount of silica-gel over the sea sand in order to get a depth of ca. 15-17
cm.
5. With the bottom valve open, gently hit the column with a rubber hose to compact the silica-
gel as much as possible. A height loss of ca. 2 cm is usual.
6. Once the silica-gel has been compacted, introduce 1 cm height of sea sand on the flat top of
the dry gel bed.
7. Place an Erlenmeyer flask under the column and open the bottom valve.
8. Slowly pour the right amount of eluant by carefully sliding it along the inner wall of the
column, just avoiding a direct hit of the solvent on the sand bed. Remember that the bottom
valve must be open, otherwise the silica-gel would crack.
41
9. Once all the eluant has been placed in the column attach the upper flow controller to the
column, secure it with a clip and connect the air pump to it.
10. Adjust the air flow with the needle valve (B) in such a way that a reasonable stream of
eluant is attained at the bottom.
11. Pass the necessary amount of eluant until the silica-gel recovers room temperature (the
silica-gel warms up with its first contact with the eluant) and has a uniform appearance.
Reuse the eluant if needed repeating steps 8 to 10.
12. When all the eluant has been passed through the silica-gel and the latter looks uniform and
is at room temperature, stop the air flow and close the bottom valve (in this order!) leaving
the eluant level just at the very top of the sea sand. Avoid the silica-gel bed to run dry
because otherwise it would crack thus loosing the necessary uniformity.
Placing the sample:
13. Dissolve the sample (ca. 300 mg) in the minimum amount of eluant (ca. 1 mL).
14. Introduce the solution by means of a Pasteur pipette into the column avoiding, as you did
before, a direct hit on the sand bed.
15. Open the bottom valve and allow some eluant to run off until its level reaches the very top
of the sea sand.
16. Using the Pasteur pipette rinse the walls of the column with small portions of eluant
avoiding as always a direct hit on the sand bed.
17. Repeat step 15.
Placing the mobile phase (eluant):
18. The solvent used to pack the column is usually reused to run the column. Prepare enough
amount of eluant (Table I) and pour it carefully into the column. Once again a direct hit of
the eluant on the sand bed must be avoided at all costs. The total volume of eluant must be
placed at once because the whole process is done in a single run. Refilling of the column at
any time during separation is highly discouraged.
Separation and analysis:
19. Get numbered 20 test tubes or Erlenmeyer flasks to collect the fractions. See Table I for
typical sizes relative to the selected column diameter.
20. Repeat steps 9 an 10.
21. Fill the 20 vessels non-stop with the approximate right amount of liquid.

42
22. Stop the eluant flow by opening the needle valve and close the bottom valve.
23. Analyze all fractions by TLC.
24. Mix the fractions with the same composition and evaporate the solvent under vacuum using
the rotary evaporator.
Report
Pre-lab questions:
1. Make a table showing the physical properties (molecular mass, m.p., b.p. solubility,
flammability [for solvents only], and toxicity/hazards) of all the reagents.
2. Explain why nitration is performed of anilide (spelled with “d”) and not on aniline (spelled
with “n”).
In-lab questions:
1. Calculate the yield of nitroanilide. Is it higher than 100%? If so, explain why.
2. Calculate the ortho:para ratio of nitroanilide after CC separation.
3. Report the melting points and compare them with literature values.
4. Report and discuss the NMR results.
Post-lab questions:
1. What product(s) could be formed if the nitration reaction temperature is allowed to raise
above 35 ºC?
2. Why nitration of aniline requires the nitrogen to be “protected” as an acetamide group?
3. What is the use of adding ammonium hydroxide after nitroacetanilide hydrolysis?
4. Write a plausible mechanism for the para-red dye formation.
5. What is the reason for the intense red color of the dye?
6. Briefly search the literature for other chromatographic techniques.
7. What is the purpose of adding solid sodium chloride to the precipitated para-red?
Please, read carefully the following annex (NMR experiment) and answer the questions you
may find at the very end of it.

43
Annex to experiment 2.-
ACETANILIDE NITRATION: A PROTON NMR (
1
H-NMR ) EXERCISE
1
H-NMR is the most powerful spectroscopic technique for structure elucidation in Organic
Chemistry.
1
H-NMR literally “sees” the hydrogen atoms of a given molecule.
Let’s analyze the structure of the starting acetanilide. We may wonder:
How many hydrogen atoms are there? A total of 9.
How many “kinds” of hydrogen atoms are there? You should distinguish 3:
those in the methyl group, that of the NH group and those on the aromatic
ring.
Do the hydrogen atoms have any symmetry relationship? Some do and
some don’t. Those three at the methyl group are equivalent. The H of NH is
singular. The two ortho hydrogen atoms are equivalent to one another; the
two meta ones are as well and the para one is singular.
In summary acetanilide has 9 H, divided into 3 kinds and 5 symmetry groups.
1
H-NMR
“sees” these latter 5 groups and gives a different signal for each one of the groups.
Figure 1 shows the
1
H-NMR of acetanilide, from just 10 mg (almost nothing!) of it in 0.6
mL of CDCl
3
(deuterated solvent at 99.9%, practically without H, in order to avoid the H from the
solvent to render too strong a signal). See the video to see how the recording of the spectrum at the
spectrometer is performed.
10 9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
3.202.401.00
Chloroform-d
No. (ppm ) Value
1 [1.88 .. 2.16] 3.205
2 [6.83 .. 7.03] 1.075
3 [7.13 .. 7.30] 2.404
4 [7.42 .. 7.63] 2.095
5 [9 .6 3 .. 1 0 .08 ] 1.000
7.6 7.5 7.4 7.3 7.2 7.1 7.0 6.9
Chemical Shift (ppm)
2.301.97 1.00
Chloroform-d
Figure 1.
1
H-NMR spectrum of starting acetanilide in CDCl
3
as solvent.
How many signals do you see? You should see five of them, just the expected ones in
terms of the analyzed symmetry groups.
NH
CH
3
O

44
The signals have a different shape, they show up in different regions of the spectrum scale
and they bear different areas (see the signal expansions). The superimposed red traces over each
signal are the corresponding “integrals” that measure the relative area of the signals.
You should get the following parameters from a
1
H-NMR spectrum:
The so-called chemical shift, i.e. where you find the signals at the given spectrum scale.
Chemical shifts are measured in parts per million (ppm). In the case of acetanilide signals
are thus observed at 2.0, 7.0, 7.3, 7.5 and 9.9 ppm.
The area or integral of each signal, expressed in relative units, assigning unity to the
smallest one. The Table on the spectrum bears all integrals. Take the closest integer
number for the integrals. You must list chemical shifts and integrals as follows: 2.0 ppm
(3H), 7.0 ppm (1H), 7.3 ppm (2H), 7.5 ppm (2H) and 9.9 ppm (1H).
The shape or multiplicity of the signal. You must end up with a list like this: 2.0 ppm (3H,
singlet), 7.0 ppm (1H, multiplet), 7.3 ppm (2H, multiplet), 7.5 ppm (2H, multiplet) y 9.9
ppm (1H, broads singlet).
These three parameters i) chemical shift, ii) integral and iii) multiplicity characterize the
1
H-NMR spectrum of any substance.
DIFFERENT SUBSTANCES ALWAYS GIVE DIFFERENT SPECTRA
Figure 2 shows the
1
H-NMR spectrum of a typical crude reaction mixture from the
nitration of acetanilide.
9 8 7 6 5 4 3 2 1 0
Chemical Shift (ppm)
Chloroform-d
8.5 8.0 7.5
Chemical Shift (ppm)
211.81 200.00 15.459.30
Chloroform-d
2.10 2.05 2.00
Chemical Shift (ppm)
292.26 32.11
No. (p p m ) Value
1 [2.04 .. 2.05] 32.111
2 [2.06 .. 2.10] 292.257
3 [7.20 .. 7.29] 15.449
4 [7.54 .. 7.62] 11.845
5 [7.62 .. 7.72] 200.001
6 [8.05 .. 8.19] 211.810
7 [8.52 .. 8.61] 9.296
Figure 2.
1
H-NMR spectrum in CDCl
3
of the crude mixture of acetanilide nitration.

45
What do you see? It’s a complex spectrum because maybe you realize we have a mixture of
two isomers, ortho (2-) and para (4-) nitroanilide in different ratio. Remember that you determined
by chromatography that the para isomer was the major component.
Let’s analyze the two isomers one at a time, once they have been separated by column
chromatography. Figures 3 and 4 contain respectively the para and ortho isomers of nitroanilide.
10 8 6 4 2 0
Chemical Shift (ppm)
3.282.172.15
Chloroform-d
No. (ppm) Value
1 [1.94 .. 2.16] 3.281
2 [7.55 .. 7.74] 2.166
3 [7.81 .. 7.98] 1.000
4 [8.02 .. 8.24] 2.152
8.5 8.0 7.5 7.0
Chemical Shift (ppm)
2.192.19 1.00
Chloroform-d
Figure 3.
1
H-NMR spectrum in CDCl
3
of pure para-nitroacetanilide.
10 8 6 4 2 0
Chemical Shift (ppm)
3.241.201.061.051.051.00
Chloroform-d
No. (pp m ) Value
1 [1.9 7 .. 2 .1 4 ] 3.238
2 [7.1 5 .. 7 .3 2 ] 1.198
3 [7.5 0 .. 7 .6 7 ] 1.063
4 [8.0 6 .. 8 .2 5 ] 1.053
5 [8.4 5 .. 8 .6 6 ] 1.050
6 [10.36 .. 10.49] 1.000
8.5 8.0 7.5
Chemical Shift (ppm)
1.171.011.001.00
Chloroform-d
Figure 4.
1
H-NMR spectrum in CDCl
3
of pure ortho-nitroacetanilide.

46
Para isomer: How many groups of hydrogen atoms do you discover? A total
of 4: methyl group (3H), NH (1H), ortho hydrogen atoms to the NH (2H) and ortho
hydrogen atoms to the NO
2
(2H). How many signals do you discern? The expected
ones: 4. In summary, you should observe the following: 2.0 ppm (3H, singlet), 7.6
ppm (2H, multiplet), 7.9 ppm (1H, broad singlet) y 8.2 ppm (2H, multiplet).
Ortho isomer: How many groups of hydrogen atoms do you discover?
A total of 6: methyl group (3H), NH (1H) and four singular aromatic
hydrogens because the ortho derivative lacks symmetry in the aromatic ring.
How many signals do you discern? The expected ones: 6. In summary, you
should observe the following: 2.0 ppm (3H, singlet), 7.3 ppm (1H, multiplet),
7.8 ppm (1H, multiplet), 8.2 ppm (1H, multiplet), 8.6 ppm (1H, multiplet)
y10.4 ppm (1H, broad singlet).
Report
1. Do you understand now the spectrum of Figure 2 recorded from the crude mixture?
2. In that mixture, can you calculate the ratio between the ortho and para isomers of
nitroanilide? Does it approximately agree with the ratio obtained from CC?
3. Are you able to detect any starting material?
4. Is it clear that NMR renders crucial information about the performed reaction? Briefly
explain.
NH
CH
3
O
NO
2
NH
CH
3
O
NO
2

47
Experiment 3.- Synthesis of Isopentyl Acetate
Overview: Preparation of Fruity Fragrances
Many of the processed foods that you buy today come with an ingredient label that lists
"artificial flavors" as one of the key ingredients. Artificial flavors are simply chemical mixtures
that mimic a natural flavor in some way. Any natural flavor is normally quite complex, with
dozens or hundreds of chemicals interacting to create the taste/smell. But it turns out that many
flavors - particularly fruit flavors - have just one or a few dominant chemical components that
carry the bulk of the taste/smell signal. Many of these chemicals are called esters (an ester is a
functional group that can be formally derived from the reaction of a carboxylic acid and an alcohol
with the concomitant loss of one water molecule). For example, the ester called octyl acetate
(CH
3
COOC
8
H
17
) is a fundamental component in orange flavor. The ester called n-propyl acetate
(CH
3
COOC
3
H
7
) is a pear aroma component. The ester called isoamyl acetate (CH
3
COOC
5
H
11
) is
a fundamental component of banana flavor (incidentally, isoamyl acetate is also the alarm
pheromone of the honeybee and it serves to attract other bees and provoke them to sting!). If you
add these esters to a product, the product will taste, to some degree, like orange, pear or banana,
respectively.
In this experiment you will prepare isoamyl acetate (also known as isopentyl acetate or
banana oil) from acetic acid and isoamyl (isopentyl) alcohol.
You will perform this synthesis at the microscale level by the Fischer esterification
method:
Carboxylic esters are carboxylic acid derivatives. They can be obtained by heating a
carboxylic acid with an alcohol in the presence of catalytic amounts of mineral acids such as
sulfuric or hydrochloric acids. This reaction, known as the Fischer esterification, is a reversible
reaction and therefore, its application in the preparation of esters is limited by the equilibrium
constant, K
e
.
K
e
=
[ester][water]
[acid][alcohol]
The yield can be considerably increased by using a large excess of one of the reactants
(which can be recovered at the end of the reaction) or by constantly removing the products from
the reaction mixture. Both approaches may be used in the synthesis of esters. In this experiment
you will use an excess amount of acetic acid.

48
To separate the ester from the unreacted carboxylic acid, you will use acid-base extraction
with an aqueous sodium bicarbonate solution. Carboxylic acids with six or more carbons are
sparingly soluble in water but soluble in organic solvents. Contrarily, sodium salts of carboxylic
acids are very soluble in water and insoluble in most organic solvents. You will take advantage of
the differential solubility of ionic and nonionic compounds in water and organic solvents to
separate organic mixtures. In this experiment no organic solvent is necessary for the acid-base
extraction because isoamyl acetate is liquid and separate from the aqueous solution as an
immiscible layer.
You will purify isoamyl acetate by microscale distillation.
Safety First
Concentrated sulfuric acid is very corrosive. It may cause burns. Avoid
skin contact.
Acetic acid is corrosive and flammable.
Isoamyl alcohol is an irritant and has a somewhat unpleasant smell.
Experimental Procedure
Materials
Isoamyl alcohol (3-methyl-1-butanol) 5.45 mL, 4.4 g (50 mmol) Irritant
Glacial acetic acid 11.5 mL, 12 g (200 mmol) Flammable, corrosive
Concentrated sulfuric acid 10 drops
Corrosive
5% Sodium bicarbonate solution
15% Sodium chloride solution
1. Weight 50 mmoles of isoamyl and add 200 mmoles of
glacial acetic acid in a pre-tared 50-mL round-bottom flask. Use
a Pasteur pipet to transfer the liquids. Add 10 drops of
concentrated sulfuric acid.
2. Add a magnetic stir bar and attach a water-jacketed
condenser (Figure 1). Reflux for one hour by heating in a sand
bath.
3. Remove from heat and allow the mixture to cool to
ambient temperature. Disassemble the apparatus and transfer
the liquid to a 100 mL separating funnel.
Figure 1
Sand bath

49
4. Rinse the round-bottom flask with 10 mL of 5% sodium bicarbonate in 15% sodium
chloride solution. Slowly transfer the rinse to the separating funnel. Stir the mixture with a
microspatula until gas evolution (carbon dioxide) has subsided.
5. Cap the funnel and invert it several times to mix the layers. Frequently vent the system
to release the pressure by momentarily unscrewing the cap. Let the system settle for about 10
minutes.
6. Transfer the lower (aqueous) layer to a labelled Erlenmeyer flask, keep it until the end of
the experiment, and then discard it.
7. Wash the organic layer remaining in the separating funnel twice with 5 mL (each time)
of the sodium bicarbonate-sodium chloride solution. Invert and vent well in each wash.
O
O
HO
O
+
NaHCO
3
reaction at the
interface between
layers
O
O
upper
organic layer
+
Na
-
O
O
lower
aqueous layer
8. Collect the aqueous washes in the same labelled
Erlenmeyer flask mentioned in step 6 (to be discarded).
9. Transfer the organic layer into another Erlenmeyer flask.
Dry the organic layer by adding anhydrous MgSO
4
until the solid
no longer clumps together at the bottom of the Erlenmeyer flask
but runs freely in the liquid. Transfer the ester with a filter-Pasteur
pipet to a 25-mL round-bottom flask.
10. Assemble a distillation apparatus using a distillation
head, a boiling chip, a thermometer, a condenser and a distillation
adaptor. Heat the flask on a sand bath set at a temperature 40-50 ºC
above the boiling point of the ester (142 ºC; unfortunately in some instances this temperature
cannot be measured; ask your
instructor).
11. Distill the isoamyl
acetate until two or three drops of
liquid remain in the flask. Record
the temperature during the
distillation. Transfer the distillate to
a tared vial. Weigh the product and
record the yield of the reaction.
Pateur
pipet
Cotton
distillation
head
themomete
r
distillation
flas
k
condenser
receiving
flas
k
distillation
adaptor
water out
water in

50
Analysis
IR Spectrum of Isoamyl Acetate
Mass Spectrum of Isoamyl Acetate
51
Report
Pre-lab
1. Make a table with the physical properties including toxicity/hazards of reagents and products
for the synthesis of isoamyl acetate.
2. Write the chemical reaction for the Fischer esterification of isoamyl acetate. Show the
mechanism.
3. Make a flowchart for the preparation of isoamyl acetate.
4. Why do you extract the reaction mixture with 5% sodium bicarbonate plus 15% sodium
chloride solution? What role does each salt play?
5. Calculate the theoretical yield of the synthesis in mg (assume that the reaction goes to
completion).
In-lab
1. Report the observed boiling point of your ester and compare it with a literature value.
2. Calculate the percentage yield of the synthesis and discuss any source of product loss.
3. Discuss how you could increase the yield of your ester.
4. Assign as many bands as possible in the IR spectrum of the product.
5. Analyze the
1
H NMR spectrum of isoamyl acetate.
6. Analyze the mass spectrum of isoamyl acetate.
Post-lab questions
1. Using the curved arrow formalism, show a detailed, step-by-step, mechanism for the
esterification reaction of isoamyl alcohol with acetic anhydride. Why the reaction equilibrium is
shifted to the formation of isoamyl acetate?
2. In which step in the procedure is the catalytic sulfuric acid removed from the product?
3. What is the purpose of washing with aqueous sodium bicarbonate? What cautionary note or hint
should be included in this step?
4. What is the purpose of having sodium chloride in the aqueous solution?
5. Imagine that you are at Boulder (Colorado) and you have been given an unknown compound
and only told that it is an ester. You determine the boiling point by simple distillation to be
133°. In Boulder, the atmospheric pressure is 625 mm. What is the corrected boiling point of
the compound? Look in the table of physical data and determine which ester it might be.
6. Which of the following compound pairs could be separated by simple distillation?
- acetone and aniline
- butyl acetate and butanol
- cyclohexane and cyclohexanol
- hexanes and toluene

52
7. Refer to the figure below. If the mole fraction of B in the liquid is 0.8, what is the composition
(roughly) of B in the vapor above the liquid? What will be the reading on the thermometer at
this point?
8. Eugenol (clove oil, see chemical formula below) has a reported boiling point of 255 °C at 760
mmHg. When determining the value of a boiling point at the greatly reduced pressures used in
vacuum distillations, you must use a nomography chart rather than the boiling point correction
formulas. Use the nomogram given below (page 40) to determine the boiling point under:
(a) water aspirator vacuum, ca. 25 torr
(b) mechanical pump vacuum, ca. 0.05 torr
What kind of vacuum system would be more appropriate?
Eugenol

53
